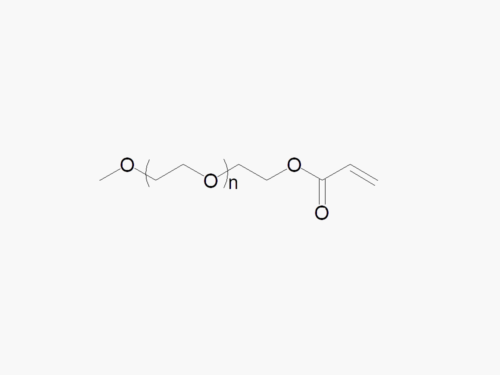
PEG products with additional MW may be made to order, please contact us for details
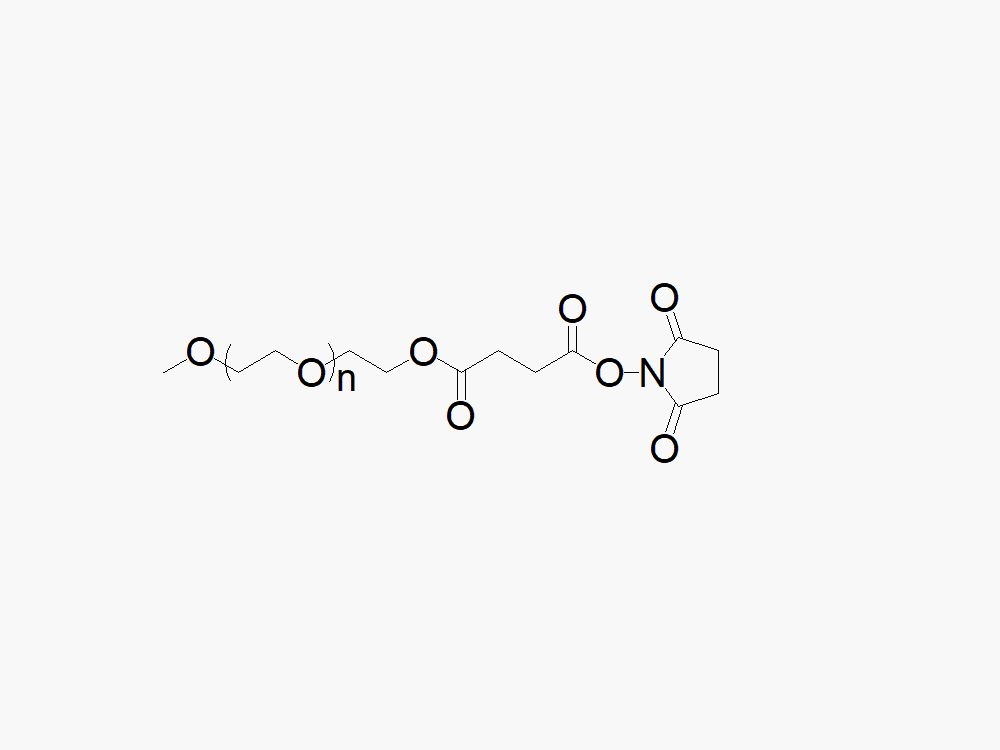
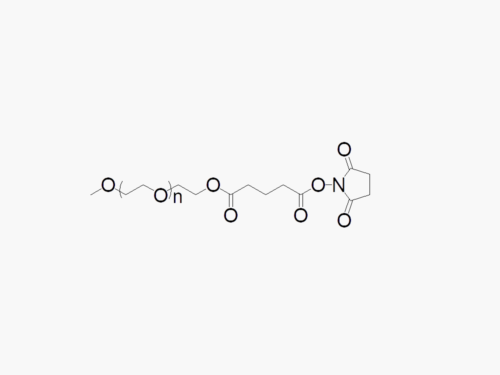
Methoxy PEG Succinimidyl Succinate
$80.00 – $3,200.00
Description
Methoxy PEG Succinimidyl Succinate with superior quality specification of ≥95% Substitution.
JenKem Technology’s Methoxy PEG Succinimidyl Succinate (Methoxy PEG SS) is a high quality amine reactive NHS PEG product with degradable cleavable ester linker between the PEG polymeric chain and the NHS ester. Linear NHS PEGs with Cleavable Linker (degradable PEGs) are especially employed in controlled release drug development applications. Methoxy PEG Succinimidyl Succinate reacts with the amine group of lysine(s), or amines such as on the active ingredients of Adagen®, Peg-ademase, or PEG-adenosine deaminase, PEG aspargase, PEG-L-asparaginase, or related biosimilars, at pH 7-8, while the ester linkage is cleaved under regular ester cleaving reaction conditions. JenKem Technology offers Methoxy PEG SS with MW 5000 Da (M-SS-5000) and MW 20000 Da (M-SS-20K), in 1g and 10g packing sizes.
Different MW of Methoxy PEG Succinimidyl Succinate products may be available by custom synthesis, please email us at tech@jenkemusa.com for details on custom PEGs. Other linear NHS esters are available – please select other cleavable linker NHS PEGs; or stable linker NHS PEGs. JenKem Technology provides repackaging services for an additional fee, please contact us if you require a different package size than our catalog selection.
Bulk PEGs and GMP grade PEGs are made-to-order. Please contact us for bulk pricing.
Click here to download the MSDS
References:
- Narvaez-Ortiz, HY, et al., Unconcerted conformational changes in Arp2/3 complex integrate multiple activating signals to assemble functional actin networks. Current Biology. 2022.
- Xiong, Q., et al., Facile fabrication of reduction-responsive supramolecular nanoassemblies for co-delivery of doxorubicin and sorafenib towards hepatoma cells, Frontiers in pharmacology, 2018, 9, p.61.
- Mastorakos, P., et al., Biodegradable brain-penetrating DNA nanocomplexes and their use to treat malignant brain tumors, Journal of Controlled Release, 2017, 262, P. 37-46.
- Mastorakos, P., et al., Biodegradable DNA Nanoparticles that Provide Widespread Gene Delivery in the Brain, Small, 2015.
- Jain, S., et al., Combinatorial bio-conjugation of gemcitabine and curcumin enables dual drug delivery with synergistic anticancer efficacy and reduced toxicity, RSC Adv., 2014, 4, 29193-29201.
- Tesfay, M.Z., et al., PEGylation of vesicular stomatitis virus extends virus persistence in blood circulation of passively immunized mice. J Virol, 2013, 87(7): p. 3752-9.
- Helgeson, L.A., et al, Mechanism of synergistic activation of Arp2/3 complex by cortactin and N-WASP, eLife, 2013, 2:e00884.
- Depp, V., et al., Native protein-initiated ATRP: A viable and potentially superior alternative to PEGylation for stabilizing biologics, Acta Biomaterialia, 5(2), 2009, p: 560-569.
Founded in 2001 by experts in PEG synthesis and PEGylation, JenKem Technology specializes exclusively in the development and manufacturing of high quality polyethylene glycol (PEG) products and derivatives, and related custom synthesis and PEGylation services. JenKem Technology is ISO 9001 and ISO 13485 certified, and adheres to ICH Q7A guidelines for GMP manufacture. The production of JenKem® PEGs is back-integrated to in-house polymerization from ethylene oxide, enabling facile traceability for regulated customers. JenKem Technology caters to the PEGylation needs of the pharmaceutical, biotechnology, medical device and diagnostics, and emerging chemical specialty markets, from laboratory scale through large commercial scale.