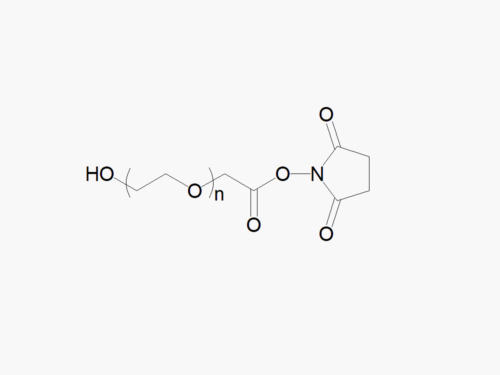
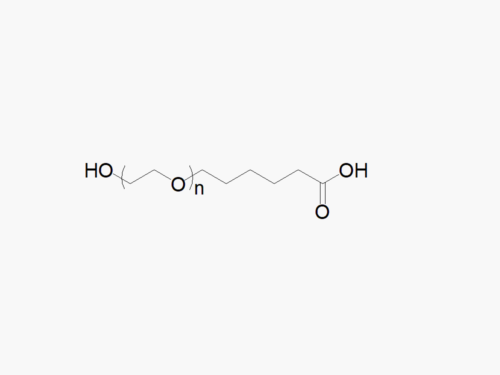
Description
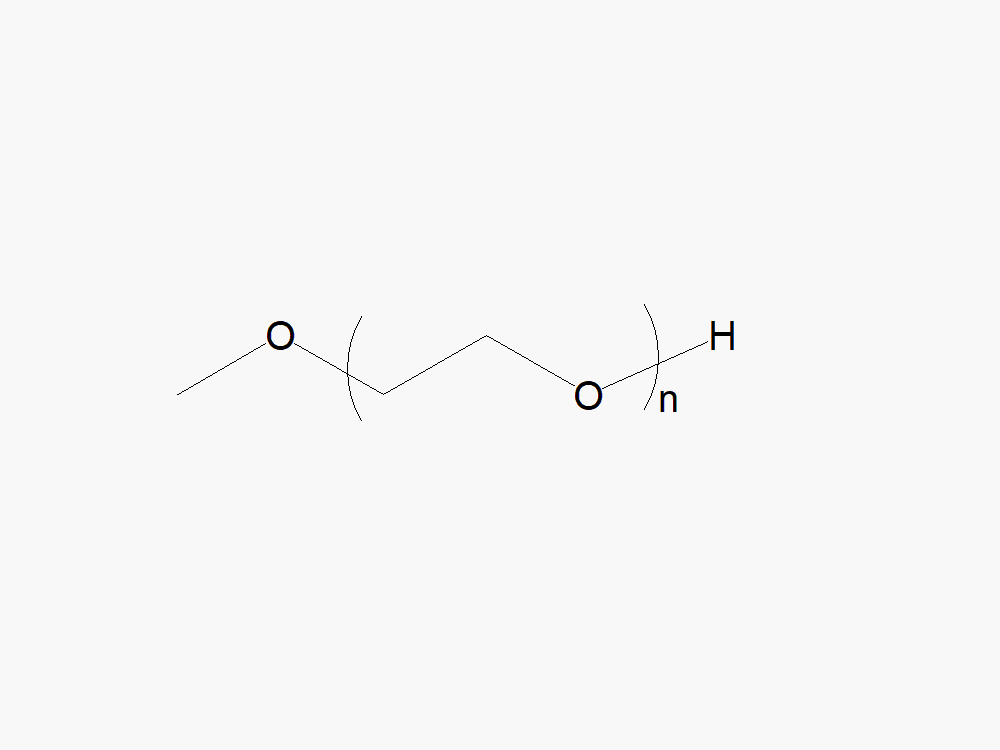
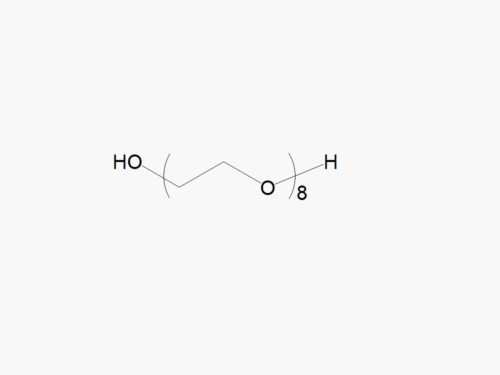
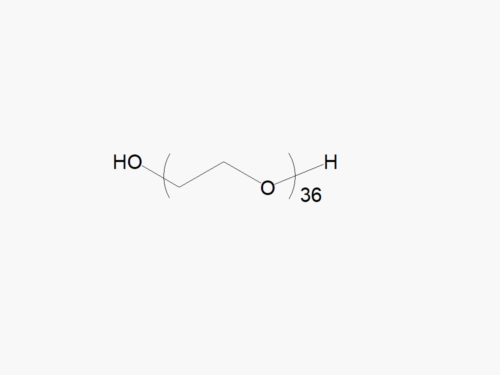
JenKem Technology’s Methoxy PEG GPC Standards kit (STDMPEG) includes 200 mg each of M-PEG-2000, M-PEG-5000, M-PEG-10K, M-PEG-20K, and M-PEG-40K (MW 2kDa, 5kDa, 10kDa, 20kDa and 40kDa, respectively).
JenKem Technology’s PEG GPC calibration standards have narrower polydispersity and more accurate molecular weights compared to other PEG standards on the market. The molecular weights of JenKem PEG standards are determined by MALDI-TOF. The polydispersity of JenKem PEG standards is determined by GPC.
Gel permeation chromatography (GPC) is a type of size exclusion chromatography. GPC separates mixture components based on molecule size, and it can be used to determine the number average molecular weight (Mn), weight average molecular weight (Mw), and peak average molecular weight of polymers. The quality of the PEG standards is very important for GPC characterization of PEGs and PEGylated products. GPC is commonly used to determine the polydispersity for polymers, which is the weight average molecular weight (Mw) over number average molecular weight (Mn).
The quality of the PEG standards is very important for GPC characterization of PEGs and PEGylated products. GPC is commonly used to determine the polydispersity for polymers, which is the weight average molecular weight (Mw) over number average molecular weight (Mn).
Click here to download the MSDS
References:
1. Abolmaali, S.S., et al., Nano-hydrogels of methoxy polyethylene glycol-grafted branched polyethyleneimine via biodegradable cross-linking of Zn2+-ionomer micelle template, Journal of nanoparticle research, 2013, 15.12, 1-21.
Founded in 2001 by experts in PEG synthesis and PEGylation, JenKem Technology specializes exclusively in the development and manufacturing of high quality polyethylene glycol (PEG) products and derivatives, and related custom synthesis and PEGylation services. JenKem Technology is ISO 9001 and ISO 13485 certified, and adheres to ICH Q7A guidelines for GMP manufacture. The production of JenKem® PEGs is back-integrated to in-house polymerization from ethylene oxide, enabling facile traceability for regulated customers. JenKem Technology caters to the PEGylation needs of the pharmaceutical, biotechnology, medical device and diagnostics, and emerging chemical specialty markets, from laboratory scale through large commercial scale.