PEG products with additional MW may be made to order, please contact us for details
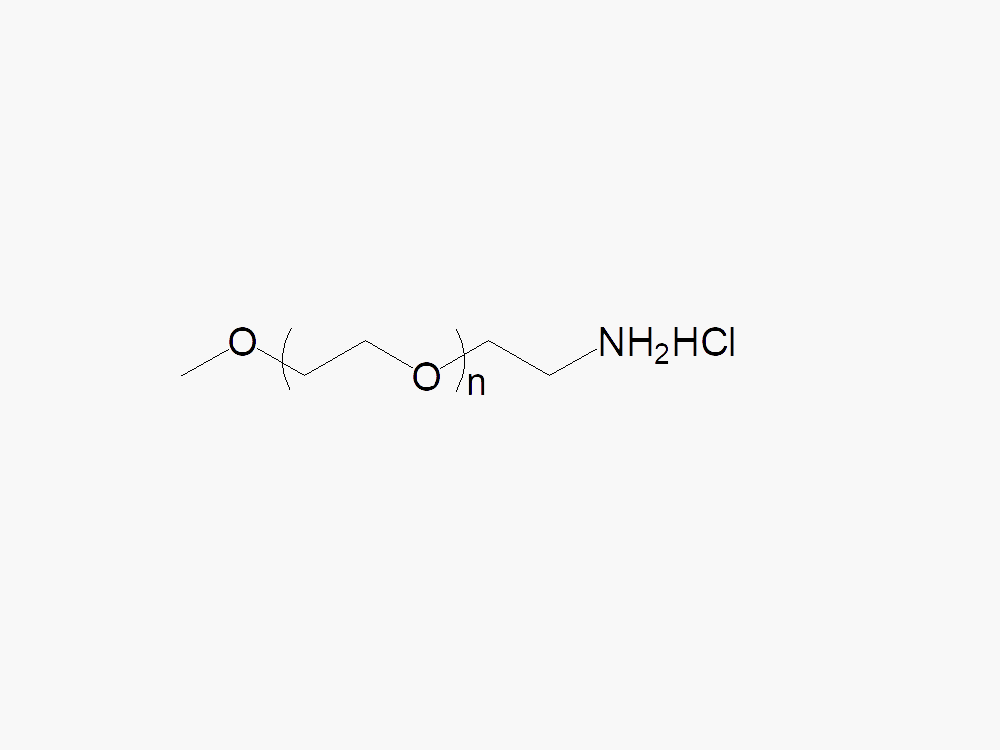
Methoxy PEG Amine, HCl Salt
$50.00 – $640.00
Description
Methoxy PEG Amine, HCl Salt, with superior quality specification of ≥95% Substitution.
Linear Amine PEGs (Methoxy PEG Amine) from JenKem Technology are high quality PEG reagents that attach via stable linkages, such as amide, urethane, urea, secondary amine. The HCl salt form provides stability for the solid form of M-PEG-NH2. JenKem Technology offers Methoxy PEG Amine HCl with MW 2kDa (M-NH2-2000), MW 3kDa (M-NH2-3000), MW 5kDa (M-NH2-5000), MW 10kDa (M-NH2-10K), MW 20kDa (M-NH2-20K), MW 30 kDa (M-NH2-30K), and MW 40kDa (M-NH2-40K), in 1g and 10g packing sizes.
Different MW of Methoxy PEG Amine products may be available by custom synthesis, please email us at tech@jenkemusa.com for details on custom PEGs. JenKem Technology provides repackaging services for an additional fee, please contact us if you require a different package size than our catalog selection.
Bulk PEGs and GMP grade PEGs are made-to-order. Please contact us for bulk pricing.
Click here to download the MSDS
References:
- Duan, H., et al., A novel electrospun nanofiber system with PEGylated paclitaxel nanocrystals enhancing the transmucus permeability and in situ retention for an efficient cervicovaginal cancer therapy, International Journal of Pharmaceutics, 650, 2024.
- Cahn, D, et al., High-Density Branched PEGylation for Nanoparticle Drug Delivery. Cellular and Molecular Bioengineering. 2022.
- Lin, M, et al., Photo-Triggered Polymeric Antimicrobial Peptide Mimics with Excellent Selectivity and Synchronizing Antifouling and Antimicrobial Hydrogels, Giant, 2022.
- Chu, Y., et al., A Neutrophil-biomimic platform for eradicating metastatic breast cancer stem-like cells by redox microenvironment modulation and hypoxia-triggered differentiation therapy, Acta Pharmaceutica Sinica B, 2022.
- Wang, M., et al., Fabrication of reversible pH-responsive aggregation-induced emission luminogens assisted by a block copolymer via a dynamic covalent bond. Polymer Chemistry. 2021, 12(19).
- Fu, D, et al., A novel redox-responsive ursolic acid polymeric prodrug delivery system for osteosarcoma therapy. Drug Delivery. 2021, 28(1):195-205.
- Hao, K, et al., Covalent organic framework nanoparticles for anti-tumor gene therapy. Science China Chemistry. 2021, 1-7.
Sheng, Q, et al., Comprehensively enhanced delivery cascade by transformable beaded nanofibrils for pancreatic cancer therapy. Nanoscale. 2021. - Deng, M, et al., pH-Triggered Copper-Free Click Reaction-Mediated Micelle Aggregation for Enhanced Tumor Retention and Elevated Immuno–Chemotherapy against Melanoma. ACS Applied Materials & Interfaces. 2021, 13(15):18033-46.
- Zhang, X, A High‐Resolution Ternary Model Demonstrates How PEGylated 2D Nanomaterial Stimulates Integrin αvβ8 on Cell Membrane. Advanced Science. 2021, 2004506.
- Xia, C., et al., Redox-responsive nanoassembly restrained myeloid-derived suppressor cells recruitment through autophagy-involved lactate dehydrogenase A silencing for enhanced cancer immunochemotherapy, Journal of Controlled Release, 2021, V. 335, P. 557-574.
- Lv, F., et al., Enhanced mucosal penetration and efficient inhibition efficacy against cervical cancer of PEGylated docetaxel nanocrystals by TAT modification, Journal of Controlled Release, 2021, V. 336, P. 572-582.
- Babity, S., et al., Data on the removal of peroxides from functionalized polyethylene glycol (PEG) and effects on the stability and sensitivity of resulting PEGylated conjugates, Data in Brief, 2020:106258.
- Li, Z., et al., Self-sterilizing diblock polycation-enhanced polyamidoxime shape-stable blow-spun nanofibers for high-performance uranium capture from seawater, Chemical Engineering Journal, 2020, V. 390.
- Sun, T., et al., Co-delivery of Cu(I) chelator and chemotherapeutics as a new strategy for tumor theranostic, Journal of Controlled Release, 2020, V. 321, P. 483-496.
- Luo, S., et al., Site specific PEGylation of β-lactoglobulin at glutamine residues and its influence on conformation and antigenicity, Food Research International, 2019, 123, p. 623-630.
- Oddone, N., et al., ROS-responsive “smart” polymeric conjugate: Synthesis, characterization and proof-of-concept study, International Journal of Pharmaceutics, 2019, 570.
- Zhang, X., et al., Inhibiting PI3 kinase-γ in both myeloid and plasma cells remodels the suppressive tumor microenvironment in desmoplastic tumors, Journal of Controlled Release, 2019, 309, p. 173-180.
- Gao, M., et al., Triggered ferroptotic polymer micelles for reversing multidrug resistance to chemotherapy, Biomaterials, 2019, 223.
- Zhang, L., et al., Highly permeable thin-film nanocomposite membranes embedded with PDA/PEG nanocapsules as water transport channels, Journal of Membrane Science, 2019, V. 586, P. 115-121.
- Doan, T.N., et al., Endothelin-1 Inhibits Size Dependent Lymphatic Clearance of PEG-Based Conjugates After Intra-Articular Injection into the Rat Knee, Acta biomaterialia, 2019.
- Xia, S., et al., Role of poly (ethylene glycol) grafted silica nanoparticle shape in toughened PLA-matrix nanocomposites, Composites Part B: Engineering, 2019.
- Muddineti, O.S., et al., Vitamin-E/lipid based PEGylated polymeric micellar doxorubicin to sensitize doxorubicin-resistant cells towards treatment, Reactive and Functional Polymers, 2019, 134:49-57.
- Tang, X., et al., PD-L1 knockdown via hybrid micelle promotes paclitaxel induced Cancer-Immunity Cycle for melanoma treatment. European Journal of Pharmaceutical Sciences, 2019, 127:161-74.
- Chong, Y.K., et al., Miktoarm star polymers nanocarrier: synthesis, characterisation, and in-vitro drug release study, Journal of Polymer Research, 2019, 26(3):79.
- Nie, W.C., et al., Orthogonal construction of dual dynamic covalent linkages toward an “AND” logic-gate acid-/salt-responsive block copolymer, Polymer, 2018, V. 159, P. 32-38.
- Lang, T., et al., Tumor microenvironment-responsive docetaxel-loaded micelle combats metastatic breast cancer, Science Bulletin, 2018.
- Muddineti, O.S., et al., Cholesterol and vitamin E-conjugated PEGylated polymeric micelles for efficient delivery and enhanced anticancer activity of curcumin: evaluation in 2D monolayers and 3D spheroids, Artificial cells, nanomedicine, and biotechnology, 2018, p.1-14.
- Yu, S., et al., A density gradient of VAPG peptides on a cell-resisting surface achieves selective adhesion and directional migration of smooth muscle cells over fibroblasts. Acta biomaterialia, 2018, 72, p.70-81.
- Yang, J., et al., A positron emission tomography image-guidable unimolecular micelle nanoplatform for cancer theranostic applications, Acta Biomaterialia, 2018, V. 79, P. 306-316.
- Gao, M., et al., All-active antitumor micelles via triggered lipid peroxidation, Journal of Controlled Release, 2018, V. 286, P. 381-393.
- Jia, H.R., et al., Plasma membrane-anchorable photosensitizing nanomicelles for lipid raft-responsive and light-controllable intracellular drug delivery, Journal of Controlled Release, 2018, V. 286, P. 103-113.
- Muddineti, O.S., et al., Transferrin-Modified Vitamin-E/Lipid Based Polymeric Micelles for Improved Tumor Targeting and Anticancer Effect of Curcumin, Pharmaceutical research, 2018, 35(5):97.
- Zhang, X., et al., Pharmaceutical micelles featured with singlet oxygen-responsive cargo release and mitochondrial targeting for enhanced photodynamic therapy, Nanotechnology, 2018, 29(25), p. 255101.
- Gao, D., et al., Polymeric micelles encapsulating pH-responsive doxorubicin prodrug and glutathione-activated zinc (II) phthalocyanine for combined chemotherapy and photodynamic therapy, Journal of Controlled Release, 2018.
- Shu, F., et al., Fabrication of a hyaluronic acid conjugated metal organic framework for targeted drug delivery and magnetic resonance imaging, RSC Advances, 2018, 8(12), p.6581-6589.
- Wang, L., et al., Delivery of radix ophiopogonis polysaccharide via sucrose acetateisobutyrate-based in situ forming systems alone or combined with itsmono-PEGylation, Drug delivery, 2018, 25(1), p.267-277.
- Wojnilowicz, M., et al., Glycogen-nucleic acid constructs for gene silencing in multicellular tumor spheroids, Biomaterials, 2018.
- Khondee, S., et al., Doxorubicin-Loaded Micelle Targeting MUC1: A Potential Therapeutic for MUC1 Triple Negative Breast Cancer Treatment, Current drug delivery, 2018, 15(3), p.406-416.
- Sun, T., et al., A targeting theranostics nanomedicine as an alternative approach for hyperthermia perfusion, Biomaterials, 2018.
- Pan, C., et al., Theranostic pH-sensitive nanoparticles for highly efficient targeted delivery of doxorubicin for breast tumor treatment. International Journal of Nanomedicine, 2018, 13, p.1119.
- Wang, X., et al., Polycation-telodendrimer nanocomplexes for intracellular protein delivery. Colloids and Surfaces B: Biointerfaces, 2018, 162, p.405-414.
- Wu, T., et al., Synthesis and characterization of poly (ethylene oxide)/polylactide/polylysine tri‐arm star copolymers for gene delivery, Journal of Polymer Science Part A: Polymer Chemistry, 2018, 56(6), p.635-644.
- Li, H., et al., Lactoferrin functionalized PEG-PLGA nanoparticles of shikonin for brain targeting therapy of glioma, International journal of biological macromolecules, 2018, 107, p.204-211.
- Wang, X., et al., Polycation-telodendrimer nanocomplexes for intracellular protein delivery, Colloids and Surfaces B: Biointerfaces, 2018, V. 162, p. 405-414.
- Balasso, A., et al., Re-programming pullulan for targeting and controlled release of doxorubicin to the hepatocellular carcinoma cells, European Journal of Pharmaceutical Sciences, 2017.
- Jalalvandi, E., et al., Schiff-base based hydrogels as degradable platforms for hydrophobic drug delivery, European Polymer Journal, 2017, 90:13-24.
- Etienne, G., et al., Influence of Fluorinated Surfactant Composition on the Stability of Emulsion Drops, Macromolecular Chemistry and Physics, 2017, 218(2).
- Clawson, G.A., et al., A Cholecystokinin B Receptor-Specific DNA Aptamer for Targeting Pancreatic Ductal Adenocarcinoma, Nucleic acid therapeutics, 2017, 27(1):23-35.
- Giraldo, J.A., et al., The impact of cell surface PEGylation and short-course immunotherapy on islet graft survival in an allogeneic murine model, Acta Biomaterialia, 2017, 49:272-83.
- Yang, Q., Pretargeting with bispecific fusion proteins facilitates delivery of nanoparticles to tumor cells with distinct surface antigens, Journal of Controlled Release, 2017.
- Khandhar, A.P., et al.,. Evaluation of PEG-coated iron oxide nanoparticles as blood pool tracers for preclinical magnetic particle imaging. Nanoscale, 2017.
- Huo, M., et al., Tumor-targeted delivery of sunitinib base enhances vaccine therapy for advanced melanoma by remodeling the tumor microenvironment, Journal of Controlled Release, 2017, V. 245, P. 81-94.
- Chen, G., et al., Tumor-targeted pH/redox dual-sensitive unimolecular nanoparticles for efficient siRNA delivery. Journal of Controlled Release, 2017.
- Jia, H.R., et al., Cholesterol-Assisted Bacterial Cell Surface Engineering for Photodynamic Inactivation of Gram-Positive and Gram-Negative Bacteria, ACS Applied Materials & Interfaces, 2017.
- Leiro, V., et al., Biodegradable PEG–dendritic block copolymers: synthesis and biofunctionality assessment as vectors of siRNA, Journal of Materials Chemistry B, 2017.
- Muddineti, O.S., et al., d-α-Tocopheryl Succinate/Phosphatidyl Ethanolamine Conjugated Amphiphilic Polymer-Based Nanomicellar System for the Efficient Delivery of Curcumin and To Overcome Multiple Drug Resistance in Cancer, ACS Applied Materials & Interfaces, 2017.
- Dong, Z., et al., Cell Isolation and Recovery Using Hollow Glass Microspheres Coated with Nanolayered Films for Applications in Resource-Limited Settings, ACS Applied Materials & Interfaces, 2017.
- Du, B., et al., Positively charged graphene/Fe3O4/polyethylenimine with enhanced drug loading and cellular uptake for magnetic resonance imaging and magnet-responsive cancer therapy, Nano Research, 2017, 1-6.
- McCallen, J.D., et al., Cross-reactivity of select PEG-binding antibodies to other polymers containing CH2-CH2-O backbone, ACS Biomaterials Science & Engineering, 2017.
- Zhao, Y., et al., Near-infrared quantum dot and 89Zr dual-labeled nanoparticles for in vivo Cerenkov imaging. Bioconjugate chemistry, 2017, 28(2):600-8.
- Peng, J., et al., “One-for-All”-Type, Biodegradable Prussian Blue/Manganese Dioxide Hybrid Nanocrystal for Trimodal Imaging-Guided Photothermal Therapy and Oxygen Regulation of Breast Cancer. ACS Applied Materials & Interfaces, 2017, 9(16):13875-86.
- Guo, Z., pH Triggered Size Increasing Gene Carrier for Efficient Tumor Accumulation and Excellent Anti-tumor Effect. ACS Applied Materials & Interfaces, 2017.
- Goh, S.C.M, Universal Aqueous-Based Antifouling Coatings For Multi-Material Devices, McMaster University, 2017.
- Wang, Y., et al., Carboplatin‐Complexed and cRGD‐Conjugated Unimolecular Nanoparticles for Targeted Ovarian Cancer Therapy, Macromolecular bioscience, 2017, 17(5).
- Guo, D., et al., Riboflavin-containing telodendrimer nanocarriers for efficient doxorubicin delivery: High loading capacity, increased stability, and improved anticancer efficacy, Biomaterials, 2017.
- Goor, O., et al., Introduction of anti-fouling coatings at the surface of supramolecular elastomeric materials via post-modification of reactive supramolecular additives, Polymer Chemistry, 2017.
- Parker, C. L., et al., Multivalent interactions between streptavidin-based pretargeting fusion proteins and cell receptors impede efficient internalization of biotinylated nanoparticles, Acta Biomaterialia, 2017, V. 63, P. 181-189.
- Soni, K.S., et al., Tuning polypeptide-based micellar carrier for efficient combination therapy of ErbB2-positive breast cancer, Journal of Controlled Release, 2017, V. 264, P. 276-287.
- Ni, Y., et al., Two-Dimensional Supramolecular Assemblies from pH-Responsive Poly (ethyl glycol)-b-poly (l-glutamic acid)-b-poly (N-octylglycine) Triblock Copolymer, Biomacromolecules, 2017, 18(10):3367-74.
- Cheng, X. Q., et al., Tailoring nanofiltration membrane performance for highly-efficient antibiotics removal by mussel-inspired modification, Journal of Membrane Science, 2016, 499, p. 326-334.
- Zhang, Y., et al., Folic acid-targeted disulfide-based cross-linking micelle for enhanced drug encapsulation stability and site-specific drug delivery against tumors. International journal of nanomedicine, 2016, 11:1119.
- Cheung, K., et al., Proteomic Analysis of the Mammalian Katanin Family of Microtubule-severing Enzymes Defines KATNBL1 as a Regulator of Mammalian Katanin Microtubule-severing, Molecular & Cellular Proteomics, 2016.
- Li, Q., et al., A new biosafe reactive oxygen species (ROS)-responsive nanoplatform for drug delivery. RSC Advances, 2016, 6(45):38984-9.
- Wang, X., et al., Affinity-Controlled Protein Encapsulation into Sub-30 nm Telodendrimer Nanocarriers by Multivalent and Synergistic Interactions, Biomaterials, 2016.
- Cheung, K., et al., Proteomic Analysis of the Mammalian Katanin Family of Microtubule-severing Enzymes Defines Katanin p80 subunit B-like 1 (KATNBL1) as a Regulator of Mammalian Katanin Microtubule-severing. Molecular & Cellular Proteomics, 2016, 15(5):1658-69.
- Guo, M., et al., Functionalization of Cellulose Nanocrystals with PEG-metal-chelating Diblock Copolymers via Controlled Conjugation in Aqueous Medium. Diss., 2016.
- Samaroo, K.J., et al., Binding and lubrication of biomimetic boundary lubricants on articular cartilage, Journal of Orthopaedic Research, 2016.
- Wen, H., et al., Assembly and Reassembly of Polyelectrolyte Complex Formed by Poly (ethylene glycol)-block-poly (glutamate sodium) and S5R4 Peptide, Macromolecules, 2016.
- Niu, Y., et al., Functional computer-to-plate near-infrared absorbers as highly efficient photoacoustic dyes, Acta Biomaterialia, 2016.
- Shi, C., et al., A drug-specific nanocarrier design for efficient anticancer therapy, Nature Communications, 2015, 6: 7449.
- Uritu, C.M., et al., Hybrid fullerene conjugates as vectors for DNA cell-delivery, J. Mater. Chem. B, 2015, 3, 2433-2446.
- Wibowo, S. H., et al., Functional and Well-Defined β-Sheet-Assembled Porous Spherical Shells by Surface-Guided Peptide Formation, Advanced Functional Materials, 2015.
- Ran, R., et al., PEGylated Hyaluronic Acid-Modified Delivery System with Anti-γ-Glutamylcyclotransferase siRNA for Drug-Resistant MCF-7 Breast Cancer Therapy, Journal of Pharmaceutical Sciences, 2015, 104(2): 476-484.
- Yao, C., et al., Increased cardiac distribution of mono-PEGylated Radix Ophiopogonis polysaccharide in both myocardial infarction and ischemia/reperfusion rats, Int J Nanomedicine, 2015, 10:409-18.
- Cheng, X.Q., et al., Nanofiltration membrane achieving dual resistance to fouling and chlorine for “green” separation of antibiotics, Journal of Membrane Science, 2015, V. 493, P. 156-166.
- Tian, Y., et al., Light- and pH-activated intracellular drug release from polymeric mesoporous silica nanoparticles, Colloids and Surfaces B: Biointerfaces, 2015, V. 134, P. 147-155.
- Alibolandi, M., et al., The chemotherapeutic potential of doxorubicin-loaded PEG-b-PLGA nanopolymersomes in mouse breast cancer model, European Journal of Pharmaceutics and Biopharmaceutics, 2015, V. 94, P. 21-531.
- Xu, G., et al., Functional-segregated coumarin-containing telodendrimer nanocarriers for efficient delivery of SN-38 for colon cancer treatment, Acta Biomaterialia, 2015, V. 21, P. 85-98.
- Quan, X.Q., et al., Synthesis of PEGylated hyaluronic acid for loading dichloro(1,2-diaminocyclohexane)platinum(II) (DACHPt) in nanoparticles for cancer treatment, Chinese Chemical Letters, 2015, 26:6, P. 695-699.
- Cai, L., et al., Telodendrimer nanocarrier for co-delivery of paclitaxel and cisplatin: A synergistic combination nanotherapy for ovarian cancer treatment, Biomaterials, 2015, 37, p: 456-468.
- Feng, G., et al, Gene therapy for nucleus pulposus regeneration by heme oxygenase-1 plasmid DNA carried by mixed polyplex micelles with thermo-responsive heterogeneous coronas, Biomaterials, 2015, 52, p: 1-13.
- Dai, X., et al., Combined Delivery of Let-7b MicroRNA and Paclitaxel via Biodegradable Nanoassemblies for the Treatment of KRAS Mutant Cancer, Molecular Pharmaceutics Article, 2015.
- Zhang, X., et al., Tunable pH-Responsive Polymeric Micelle for Cancer Treatment, ACS Macto Lett., 2015, 4, pp 620–623.
- Gao, D., et al., Ultrasound-triggered phase transition cationic nanodroplets for enhanced gene delivery, ACS Appl. Mater. Interfaces, 2015.
- Chen, P., et al., pH-Responsive Chimaeric Pepsomes Based on Asymmetric Poly(ethylene glycol)-b-Poly(l-leucine)-b-Poly(l-glutamic acid) Triblock Copolymer for Efficient Loading and Active Intracellular Delivery of Doxorubicin Hydrochloride, Biomacromolecules, 2015, 16(4), p. 1322–1330.
- Tang, S., et al., Inhibition of metastasis and growth of breast cancer by pH-sensitive poly (β-amino ester) nanoparticles co-delivering two siRNA and paclitaxel, Biomaterials, 2015, 48, P. 1-15.
- Huang, W., et al., Fine-Tuning Vitamin E-Containing Telodendrimers for Efficient Delivery of Gambogic Acid in Colon Cancer Treatment, Molecular Pharmaceutics, 2015, 12 (4), 1216-1229.
- Yu, L., et al., Tuning the phase transition temperature of thermal-responsive OEGylated poly-L-glutamate via random copolymerization with L-alanine, Soft Matter, 2015, 11, 545-550.
- Wen, H., et al., Complete dissociation and reassembly behavior as studied by using poly(ethylene glycol)-block-poly(glutamate sodium) and kanamycin A, Soft Matter, 2015, 11, 1930-1936.
- Zolotarskaya, O.Y., et al. Click synthesis of a polyamidoamine dendrimer-based camptothecin prodrug, RSC Adv., 2015, 5, 58600-58608.
- Wang, L., et al., Targeting delivery of Radix Ophiopogonis polysaccharide to ischemic/reperfused rat myocardium by long-circulating macromolecular and liposomal carriers, International Journal of Nanomedicine, 2015, 10:5729-5737.
- Cheng, X.Q., et al., High flux polyethylene glycol based nanofiltration membranes for water environmental remediation, Journal of Membrane Science, 2015, 476, P. 95-104.
- Zhao, Y., et al., Nanoparticle delivery of CDDO-Me remodels the tumor microenvironment and enhances vaccine therapy for melanoma, Biomaterials, 2015, V. 68, P. 54-66.
- Xu, P., et al., Shrapnel nanoparticles loading docetaxel inhibit metastasis and growth of breast cancer, Biomaterials, 2015, V. 64, P. 10-20.
- Liu, J., et al., Integrin-targeted pH-responsive micelles for enhanced efficiency of anticancer treatment in vitro and in vivo, Nanoscale, 2015, 7, 4451-4460.
- Sun, Y., et al., Fabrication of mPEGylated graphene oxide/poly(2-dimethyl aminoethyl methacrylate) nanohybrids and their primary application for small interfering RNA delivery. J. Appl. Polym. Sci., 2015, 43303.
- Fan, W., et al., Reduction-responsive Crosslinked Micellar Nanoassemblies for Tumor-targeted Drug Delivery, Pharmaceutical Research, 2015, 32:4, p. 1325-1340.
- Deng, L., et al., A strategy for oral chemotherapy via dual pH-sensitive polyelectrolyte complex nanoparticles to achieve gastric survivability, intestinal permeability, hemodynamic stability and intracellular activity, European Journal of Pharmaceutics and Biopharmaceutics, 2015, 97A, P. 107-117.
- Ashton, A.W., et al., Role of Nongenomic Signaling Pathways Activated by Aldosterone During Cardiac Reperfusion Injury, Molecular Endocrinology, 2015, 29:8, 1144-1155 .
- Abouelmagd, S.A., et al., Low molecular weight chitosan-coated polymeric nanoparticles for sustained and pH-sensitive delivery of paclitaxel, Journal of Drug Targeting, 2015, 23:7-8.
- Luo, N., et al., Surface-Engineered Graphene Navigate Divergent Biological Outcomes toward Macrophages, ACS Applied Materials & Interfaces, 2015, 7 (9), 5239-5247.
- Zhang, K., et al., Poloxamer-based in situ hydrogels for controlled delivery of hydrophilic macromolecules after intramuscular injection in rats, Drug Delivery, 2015, 22:3.
- Xue, X., et al., Heat treatment increases the bioactivity of C-terminally PEGylated staphylokinase. Process Biochemistry, 2014, 49(7): p. 1092-1096.
- Moreno, M., et al., Delivering wasp venom for cancer therapy, Journal of Controlled Release, 2014, 182(0): p. 13-21.
- Kim, K.S., et al., A cancer-recognizable MRI contrast agents using pH-responsive polymeric micelle. Biomaterials, 2014, 35(1): p. 337-343.
- Kang, T., et al., iNGR-modified PEG-PLGA nanoparticles that recognize tumor vasculature and penetrate gliomas. Biomaterials, 2014, 35(14): p. 4319-4332.
- Bai, J., et al., Multifunctional PEG-GO/CuS nanocomposites for near-infrared chemo-photothermal therapy. Biomaterials, 2014, 35(22): p. 5805-5813.
- Guo, M., et al., Dual imaging-guided photothermal/photodynamic therapy using micelles. Biomaterials, 2014, 35(16): p. 4656-4666.
- Ma, Y., et al., Irreversible Low Critical Solution Temperature Behaviors of Thermal-responsive OEGylated Poly(l-cysteine) Containing Disulfide Bonds, Macromolecules, 2014.
- Chen, J., et al., Reducible polyamidoamine-magnetic iron oxide self-assembled nanoparticles for doxorubicin delivery. Biomaterials, 2014, 35(4): p. 1240-1248.
- Ran, R., et al., Enhanced gene delivery efficiency of cationic liposomes coated with PEGylated hyaluronic acid for anti P-glycoprotein siRNA: A potential candidate for overcoming multi-drug resistance, International Journal of Pharmaceutics, 2014, 477 (1–2), p: 590-600.
- Serano, A., Novel Polymer-Brush Based Coatings for Regulating Bioadhesion, ETH e-Collection, 2014.
- Bao, B., et al., Facile Preparation of Multicolor Polymer Nanoparticle Bioconjugates with Specific Biorecognition, ACS Applied Materials & Interfaces, 2014, 6 (14), 11129-11135.
- Yang, Q., et al., Evading Immune Cell Uptake and Clearance Requires PEG Grafting at Densities Substantially Exceeding the Minimum for Brush Conformation, Molecular Pharmaceutics, 2014, 11 (4), 1250-1258.
- Wan, Z., et al., Highly Efficient Hierarchical Micelles Integrating Photothermal Therapy and Singlet Oxygen-Synergized Chemotherapy for Cancer Eradication, Theranostics, 2014, 4(4):399-411.
- Zhang, S., et al., Supramolecular hydrogels assembled from nonionic poly(ethylene glycol)-b-polypeptide diblocks containing OEGylated poly-L-glutamate, Polym. Chem., 2014, 5, 3346-3351.
- Feng, T., et al., PEGylated poly(aspartate-g-OEI) copolymers for effective and prolonged gene transfection, J. Mater. Chem. B, 2014, 2, 2725-2732.
- Shi, X., et al., Injectable long-acting systems for Radix Ophiopogonis polysaccharide based on mono-PEGylation and in situ formation of a PLGA depot, International Journal of Nanomedicine, 2014, 9:5555-5563.
- Dao, T., et al., A new formulation of curcumin using poly (lactic-co-glycolic acid)—polyethylene glycol diblock copolymer as carrier material, Advances in Natural Sciences: Nanoscience and Nanotechnology, 2014, 5:3.
- Das, M., et al., Macromolecular Bipill of Gemcitabine and Methotrexate Facilitates Tumor-Specific Dual Drug Therapy with Higher Benefit-to-Risk Ratio, Bioconjugate Chemistry, 2014, 25 (3), 501-509.
- Shi, C., et al., A Structure–Property Relationship Study of the Well-Defined Telodendrimers to Improve Hemocompatibility of Nanocarriers for Anticancer Drug Delivery, Langmuir, 2014, 30 (23), 6878-6888.
- Yang, Z., et al., Multifunctional non-viral gene vectors with enhanced stability, improved cellular and nuclear uptake capability, and increased transfection efficiency, Nanoscale, 2014, 6, 10193-10206.
- Shao, Y., et al., Photo and Redox Dual Responsive Reversibly Cross-Linked Nanocarrier for Efficient Tumor-Targeted Drug Delivery, ACS Applied Materials & Interfaces, 2014, 6 (13), 10381-10392.
- Miller, D.J., et al., Constant flux crossflow filtration evaluation of surface-modified fouling-resistant membranes, Journal of Membrane Science, 2014, 452, P. 171-183.
- Amoozgar, Z., et al., Dual-Layer Surface Coating of PLGA-Based Nanoparticles Provides Slow-Release Drug Delivery To Achieve Metronomic Therapy in a Paclitaxel-Resistant Murine Ovarian Cancer Model, Biomacromolecules, 2014, 15 (11), 4187-4194.
- Wu, L., et al., Phenyl Amide Linker Improves the Pharmacokinetics and Pharmacodynamics of N-Terminally Mono-PEGylated Human Growth Hormone, Molecular Pharmaceutics, 2014, 11(9), 3080-3089.
- Li, F., et al., Surface modification of PES ultrafiltration membrane by polydopamine coating and poly(ethylene glycol) grafting: Morphology, stability, and anti-fouling, Desalination, 2014, 344, P. 422-430.
- Qin, Y., et al., Liposomes formulated with fMLP-modified cholesterol for enhancing drug concentration at inflammatory sites, Journal of Drug Targeting, 2014, 22:2.
- Wang, L., et al., Primary arylamine-based tyrosine-targeted protein modification, RSC Adv., 2014, 4, 39446-39452.
- Miao, L., et al., Nanoparticles with Precise Ratiometric Co-Loading and Co-Delivery of Gemcitabine Monophosphate and Cisplatin for Treatment of Bladder Cancer. Adv. Funct. Mater., 2014, 24: 6601–6611.
- Lei, Y., et al., Glutathione-sensitive RGD-poly(ethylene glycol)-SS-polyethylenimine for intracranial glioblastoma targeted gene delivery. J. Gene Med., 2013, 15: 291–305.
- Chen, J., et al., Photo-cross-linked and pH-Sensitive Biodegradable Micelles for Doxorubicin Delivery, ACS Applied Materials & Interfaces, 2013, 5 (8), 3108-3117.
- Zhang, J., et al., Long-Circulating Heparin-Functionalized Magnetic Nanoparticles for Potential Application as a Protein Drug Delivery Platform, Molecular Pharmaceutics, 2013, 10 (10), 3892-3902.
- Yang, H., et al., Micelles assembled with carbocyanine dyes for theranostic near-infrared fluorescent cancer imaging and photothermal therapy, Biomaterials, 2013, 34(36):9124-33.
- Xue, X., et al., Phenyl linker-induced dense PEG conformation improves the efficacy of C-terminally monoPEGylated staphylokinase. Biomacromolecules, 2013, 14(2):331-41.
- Blum, K.S., et al., Dynamics of lymphatic regeneration and flow patterns after lymph node dissection. Breast cancer research and treatment, 2013, 139(1):81-6.
- Nag, O.K., et al., Post-modification of preformed liposomes with novel non-phospholipid poly (ethylene glycol)-conjugated hexadecylcarbamoylmethyl hexadecanoic acid for enhanced circulation persistence in vivo. International journal of pharmaceutics, 2013, 446(1):119-29.
- Zhang, W., et al., Gene transfection efficacy and biocompatibility of polycation/DNA complexes coated with enzyme degradable PEGylated hyaluronic acid, Biomaterials, 2013, 34.27: 6495-6503.
- Cheraghipour, E., et al., PEG conjugated citrate-capped magnetite nanoparticles for biomedical applications.” Journal of Magnetism and Magnetic Materials, 2013, 328: 91-95.
- Tian, Ye, et al., Polymeric mesoporous silica nanoparticles as a pH-responsive switch to control doxorubicin intracellular delivery, Journal of Materials Chemistry B, 2013, 1.39: 5264-5272.
- Shen, J., et al., Synthesis, characterization, in vitro and in vivo evaluation of PEGylated oridonin conjugates.” International journal of pharmaceutics, 2013, 456.1: 80-86.
- Sun, G., et al., Mono-PEGylated radix ophiopogonis polysaccharide for the treatment of myocardial ischemia, European Journal of Pharmaceutical Sciences, 2013, 49.4: 629-636.
- Miller, D. J., et al., Fouling-resistant membranes for the treatment of flowback water from hydraulic shale fracturing: a pilot study, Journal of Membrane Science, 2013, 437, 265-275.
- Barrett, D. G., et al., pH‐Based Regulation of Hydrogel Mechanical Properties Through Mussel‐Inspired Chemistry and Processing, Advanced functional materials, 2013, 23.9: 1111-1119.
- Proulx, S. T., et al., Use of a PEG-conjugated bright near-infrared dye for functional imaging of rerouting of tumor lymphatic drainage after sentinel lymph node metastasis, Biomaterials, 2013, 34(21), 5128–5137.
- Huang, Y., et al., Binary and ternary complexes based on polycaprolactone-graft-poly (N, N-dimethylaminoethyl methacrylate) for targeted siRNA delivery, Biomaterials, 2012, 33:18, p. 4653-4664.
- Chen, J., et al., Reducible self-assembled micelles for enhanced intracellular delivery of doxorubicin, J. Mater. Chem., 2012, 22, 7121-7129.
- Xiao, Y., et al., Multifunctional unimolecular micelles for cancer-targeted drug delivery and positron emission tomography imaging, Biomaterials, 2012, 33(11), p: 3071-3082.
- Fonge, H., et al., Influence of formulation variables on the biodistribution of multifunctional block copolymer micelles, Journal of Controlled Release, 2012, 157:3, p. 366-374.
- Chen, H., et al., High-level production of uricase containing keto functional groups for site-specific PEGylation, Biochemical Engineering Journal, 2011, 58–59, p. 25-32.
- Qin, Y., et al., Liposome formulated with TAT-modified cholesterol for improving brain delivery and therapeutic efficacy on brain glioma in animals, International Journal of Pharmaceutics, 2011, 420:2, P. 304-312.
- Sun, G., et al., PEGylated Inulin as Long-Circulating Pharmaceutical Carrier, Journal of Biomaterials Science, Polymer Edition, 2011, 22:4-6.
- Chen, J., et al., pH and Reduction Dual-Sensitive Copolymeric Micelles for Intracellular Doxorubicin Delivery, Biomacromolecules, 2011, 12 (10), 3601-3611.
- Lin, X., et al., Comparison of tissue distribution of a PEGylated Radix Ophiopogonis polysaccharide in mice with normal and ischemic myocardium, European Journal of Pharmaceutics and Biopharmaceutics, 2011, 79:3, P. 621-626.
- Khondee, S., et al., Noncovalent PEGylation by Polyanion Complexation as a Means To Stabilize Keratinocyte Growth Factor-2 (KGF-2), Biomacromolecules, 2011, 12 (11), 3880-3894.
- Pearson, R.M., et al., Facilitated self-assembly of novel dendron-based copolymers, in Engineering in Medicine and Biology Society, EMBC, 2011, pp.8334-8336.
- Sun, J., et al., Synthesis of a novel histidine-targeting poly(ethylene glycol) and modification of lysozyme, J. Appl. Polym. Sci., 2011, 119: 2183–2188.
- Lin, X., et al., Long-circulating delivery of bioactive polysaccharide from radix ophiopogonis by PEGylation, International Journal of Nanomedicine, 2011, 6:2865-2872.
- Azemi, E., et al., The surface immobilization of the neural adhesion molecule L1 on neural probes and its effect on neuronal density and gliosis at the probe/tissue interface, Biomaterials, 2011, 32:3, P. 681-692.
- Han, H.-M., et al., Improvement of the durability of porous silicon through functionalisation for biomedical applications, Thin Solid Films, 2011, 519:10, P. 3325-3333.
- Pei, J., et al., Covalently derivatized NTA microarrays on porous silicon for multi-mode detection of His-tagged proteins, Science China Chemistry, 2011, 54:3, p. 526-535.100.
- Li, H.F., et al., Biological functionalization and patterning of porous silicon prepared by Pt-assisted chemical etching, Applied Surface Science, 2010, 256:12, P. 4048-4051.
- Fonge, H., et al., Multifunctional Block Copolymer Micelles for the Delivery of 111In to EGFR-Positive Breast Cancer Cells for Targeted Auger Electron Radiotherapy, Molecular Pharmaceutics, 2010, 7 (1), 177-186.
- Huang, C., et al., Magnetic nanoparticles for magnetic resonance imaging: modulation of macrophage uptake by controlled PEGylation of the surface coating, J. Mater. Chem., 2010, 20, 8512-8520.
- Liu, T., et al., Stability of ‘quantum dot human epidermal growth factor’ bioconjugates prepared using quantum dots synthesised in aqueous solution, Journal of Experimental Nanoscience, 2010, 5:2.
- McCloskey, B.D., et al., Influence of polydopamine deposition conditions on pure water flux and foulant adhesion resistance of reverse osmosis, ultrafiltration, and microfiltration membranes, Polymer, 2010, 51:15, P. 3472-3485.
- Lin, X., et al., Poly(ethylene glycol)-Radix Ophiopogonis polysaccharide conjugates: Preparation, characterization, pharmacokinetics and in vitro bioactivity, European Journal of Pharmaceutics and Biopharmaceutics, 2010, 76:2, P. 230-237.
- Altinoglu, E.I., et al., Near-Infrared Emitting Fluorophore-Doped Calcium Phosphate Nanoparticles for In Vivo Imaging of Human Breast Cancer, ACS Nano, 2008, 2 (10), 2075-2084.
Note: Starting July 2016, M-NH2HCl is the new item number of the product M-NH2 (MPEG Amine HCL salt).
Founded in 2001 by experts in PEG synthesis and PEGylation, JenKem Technology specializes exclusively in the development and manufacturing of high quality polyethylene glycol (PEG) products and derivatives, and related custom polymer synthesis and PEGylation services. JenKem Technology is ISO 9001 and ISO 13485 certified, and adheres to ICH Q7A guidelines for GMP manufacture. The production of JenKem® PEGs is back-integrated to in-house polymerization from ethylene oxide, enabling facile traceability for regulated customers. JenKem Technology caters to the PEGylation needs of the pharmaceutical, biotechnology, medical device and diagnostics, and emerging chemical specialty markets, from laboratory scale through large commercial scale.