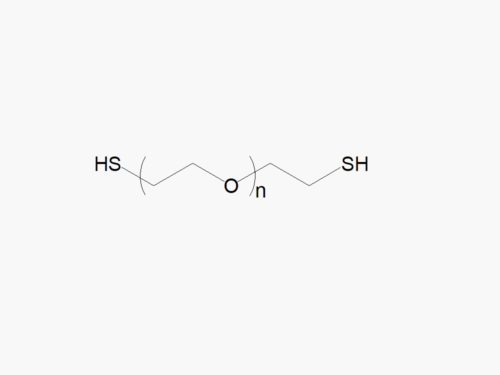
PEG products with additional MW may be made to order, please contact us for details
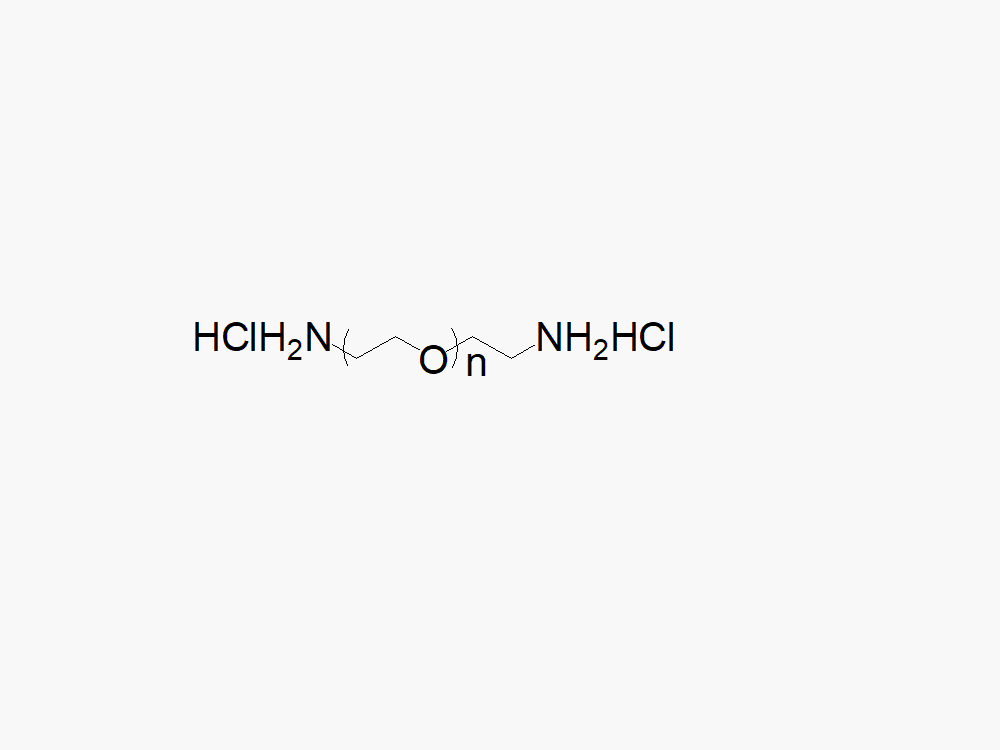
PEG (Amine)2, HCl Salt
$60.00 – $240.00
Description
PEG (Amine)2, HCl Salt product with superior quality specification of ≥95% Substitution.
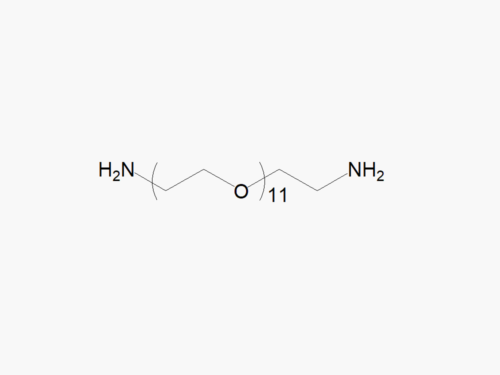
PEG (Amine)2, HCl Salt from JenKem Technology (diamine PEG) is a homobifunctional PEG employed for pegylation via stable linkages, such as amide, urethane, urea, or secondary amine. The HCl salt form provides stability for the solid form of Amine PEG Amine. Homobifunctional PEG derivatives from JenKem Technology have numerous applications as crosslinkers for PEGylation of proteins and peptides, nanoparticle and surface modifications. Conjugation with homobifunctional PEGs provides an increased drug load compared to pegylation using linear PEGs. JenKem Technology offers PEG (Amine)2, HCl Salt with MW 2,000 Da (NH2HCl-PEG2000-NH2HCl), MW 3,500 Da (NH2HCl-PEG3500-NH2HCl), MW 5,000 Da (NH2HCl-PEG5000-NH2HCl), and MW 7,500 Da (NH2HCl-PEG7500-NH2HCl), in 1g and 5g packing sizes.
Different MW of PEG diAmine may be available by custom synthesis, please email us at tech@jenkemusa.com for details on custom PEGs. JenKem Technology provides repackaging services for an additional fee, please contact us if you require a different package size than our catalog selection.
Bulk PEGs and GMP grade PEGs are made-to-order. Please contact us for bulk pricing.
Note: Starting July 2016, PEG (Amine)2, HCl Salt is the new name of the product Amine PEG Amine HCl salt (MW 2000 (NH2-PEG2000-NH2), MW 3500 (NH2-PEG3500-NH2), MW 5000 (NH2-PEG5000-NH2) and MW 7500 (NH2-PEG7500-NH2)).
References:
- Shalumon, K. T., et al., Braided suture-reinforced fibrous yarn bundles as a scaffold for tendon tissue engineering in extensor digitorum tendon repair, Chemical Engineering Journal, V. 454(3), 2023.
- Hu, Q., et al., The Promotional Effect of Hollow MnO2 with Brucea Javanica Oil Emulsion (BJOE) on Endometrial Cancer Apoptosis. BioMed Research International. 2021.
- Lu, J, et al, Dual-Labeled Graphene Quantum Dot-Based Förster Resonance Energy Transfer Nanoprobes for Single-Molecule Localization Microscopy. ACS omega. 2021, 6(13):8808-15.
- Saniee, F, et al., Glutamate-urea-based PSMA-targeted PLGA nanoparticles for prostate cancer delivery of docetaxel. Pharmaceutical Development and Technology. 2021, 26(4):381-9.
- Yu, H., et al., Polyvinylpyrrolidone functionalization induces deformable structure of graphene oxide nanosheets for lung-targeting delivery, Nano Today, 2021, V. 38.
- Liao, H. T., et al, A bioactive multi-functional heparin-grafted aligned poly(lactide-co-glycolide)/curcumin nanofiber membrane to accelerate diabetic wound healing, Materials Science and Engineering: C, 2021, V. 120.
- Yang, S.-B., et al., The colossal role of H-MnO2-PEG in ischemic stroke, Nanomedicine: Nanotechnology, Biology and Medicine, 2021, 33, 102362.
- Kawanami, T., et al., A novel diclofenac-hydrogel conjugate system for intraarticular sustained release: Development of 2-pyridylamino-substituted 1-phenylethanol (PAPE) and its derivatives as tunable traceless linkers, International Journal of Pharmaceutics, 2020, 585, 119519.
- Zhang, T., et al., Magnetic/pH dual-responsive nanocomposites loading doxorubicin hydrochloride for cancer therapy, Micro & Nano Letters, 2019.
- Goh, S.C., et al., Polydopamine-polyethylene glycol-albumin antifouling coatings on multiple substrates: variations, Journal of Materials Chemistry B, 2018.
- Kushwah, V., at al., Improved antitumor efficacy and reduced toxicity of docetaxel using anacardic acid functionalized stealth liposomes, Colloids and Surfaces B: Biointerfaces, 2018, V. 172, P. 213-223.
- Meng, Y., et al., Aminopeptidase N (CD13) targeted MR and NIRF dual-modal imaging of ovarian tumor xenograft, Materials Science and Engineering: C, 2018, V. 93, P. 968-974.
- Goor, O., et al., Introduction of anti-fouling coatings at the surface of supramolecular elastomeric materials via post-modification of reactive supramolecular additives, Polymer Chemistry, 2017.
- Goh, S.C.M, Universal Aqueous-Based Antifouling Coatings For Multi-Material Devices, McMaster University, 2017.
- Chen, X., et al., PLGA-PEG-PLGA triblock copolymeric micelles as oral drug delivery system: In vitro drug release and in vivo pharmacokinetics assessment, Journal of Colloid and Interface Science, 2017, V. 490, P. 542-552.
- Mou, J., et al., A New Green Titania with Enhanced NIR Absorption for Mitochondria-Targeted Cancer Therapy, Theranostics, 2017, 7(6):1531-1542.
- Kim, H.C., et al., Highly stable and reduction responsive micelles from a novel polymeric surfactant with a repeating disulfide-based gemini structure for efficient drug delivery, Polymer, 2017.
- Bai, J., et al., Triple-Modal Imaging of Magnetically-Targeted Nanocapsules in Solid Tumours In Vivo, Theranostics, 2016, 6(3):342-356.
- Jain, S., et al., Estradiol functionalized multi-walled carbon nanotubes as renovated strategy for efficient gene delivery, RSC Advances, 2016, 6(13):10792-801
- Mehdizadeh, M., et al., Biotin decorated PLGA nanoparticles containing SN-38 designed for cancer therapy. Artificial cells, nanomedicine, and biotechnology, 2016, 1-0.
- Bai, J., et al., Real-time monitoring of magnetic drug targeting using fibered confocal fluorescence microscopy, Journal of Controlled Release, 2016.
- Kippstein, R., et al., Passively Targeted Curcumin-Loaded PEGylated PLGA Nanocapsules for Colon Cancer Therapy In Vivo, Small, 2015, 11: 4704–4722.
- Jain, S., et al., Enhanced Antitumor Efficacy and Reduced Toxicity of Docetaxel Loaded Estradiol Functionalized Stealth Polymeric Nanoparticles, Mol. Pharmaceutics, 2015, 12 (11), pp 3871–3884.
- Chen, N., et al., Cy5.5 conjugated MnO nanoparticles for magnetic resonance/near-infrared fluorescence dual-modal imaging of brain gliomas, Journal of Colloid and Interface Science, 2015, 457, P. 27-34.
- Liu, S., et al., Meter-long multiblock copolymer microfibers via interfacial bioorthogonal polymerization, Adv. Mater., 2015.
- Zhang, T., et al., Targeted nanodiamonds as phenotype-specific photoacoustic contrast agents for breast cancer, Nanomedicine, 2015, 10:4 , P. 573-587.
- Cheng, L., et al., Construction and evaluation of PAMAM–DOX conjugates with superior tumor recognition and intracellular acid-triggered drug release properties, Colloids and Surfaces B: Biointerfaces, 2015, 136, P. 37-45.
- Li, S., et al., Targeted imaging of brain gliomas using multifunctional Fe3O4/MnO nanoparticles, RSC Adv., 2015, 5, 33639-33645.
- Chen, W., et al., Assembly of Fe3O4 nanoparticles on PEG-functionalized graphene oxide for efficient magnetic imaging and drug delivery, RSC Adv., 2015, 5, 69307-69311.
- Rubio, N., et al., Solvent-Free Click-Mechanochemistry for the Preparation of Cancer Cell Targeting Graphene Oxide, ACS Applied Materials & Interfaces, 2015, 7 (34), 18920-18923.
- Chen, H., et al., A graphene quantum dot-based FRET system for nuclear-targeted and real-time monitoring of drug delivery, Nanoscale, 2015, 7, 15477-15486.
- El-Gogary, R.I., et al., Polyethylene Glycol Conjugated Polymeric Nanocapsules for Targeted Delivery of Quercetin to Folate-Expressing Cancer Cells in Vitro and in Vivo. ACS Nano, 2014, 8(2): p. 1384-1401.
- Amoozgar, Z., et al., Dual-layer surface coating of PLGA-based nanoparticles provides slow-release drug delivery to achieve metronomic therapy in a paclitaxel-resistant murine ovarian cancer model, Biomacromolecules, 2014, 15(11):4187-94.
- Feng, T., et al., PEGylated poly(aspartate-g-OEI) copolymers for effective and prolonged gene transfection, J. Mater. Chem. B, 2014, 2, 2725-2732.
- Browning, M.B., et al., Endothelial Cell Response to Chemical, Biological, and Physical Cues in Bioactive Hydrogels, Tissue Engineering Part A, 2014, 20(23-24): 3130-3141.
- Zhang, M., Graphene Oxide Based Theranostic Platform for T1-Weighted Magnetic Resonance Imaging and Drug Delivery, ACS Appl. Mater. Interfaces, 2013, 5(24), pp 13325–13332.
- Das, M., et al., Intranuclear Drug Delivery and Effective in Vivo Cancer Therapy via Estradiol–PEG-Appended Multiwalled Carbon Nanotubes, Mol. Pharmaceutics, 2013, 10 (9), pp 3404–3416.
- Zhou, J., et al., In vivo evaluation of medical device-associated inflammation using a macrophage-specific positron emission tomography (PET) imaging probe. Bioorganic & Medicinal Chemistry Letters, 2013, 23(7): p. 2044-2047.
- Browning, M.B., Bioactive Hydrogels with Enhanced Initial and Sustained Cell Interactions, Biomacromolecules, 2013, 14(7) p: 2225–2233.
- Bagaria, H.G., et al., Adsorption of iron oxide nanoclusters stabilized with sulfonated copolymers on silica in concentrated NaCl and CaCl 2 brine, Journal of colloid and interface science, 2013, 398: 217-226.
- Zhang, T., et al., In vivo photoacoustic imaging of breast cancer tumor with HER2-targeted nanodiamonds, SPIE NanoScience+ Engineering, International Society for Optics and Photonics, 2013, pp. 881504-881504.
Founded in 2001 by experts in PEG synthesis and PEGylation, JenKem Technology specializes exclusively in the development and manufacturing of high quality polyethylene glycol (PEG) products and derivatives, and related custom synthesis and PEGylation services. JenKem Technology is ISO 9001 and ISO 13485 certified, and adheres to ICH Q7A guidelines for GMP manufacture. The production of JenKem® PEGs is back-integrated to in-house polymerization from ethylene oxide, enabling facile traceability for regulated customers. JenKem Technology caters to the PEGylation needs of the pharmaceutical, biotechnology, medical device and diagnostics, and emerging chemical specialty markets, from laboratory scale through large commercial scale.