PEG products with additional MW may be made to order, please contact us for details
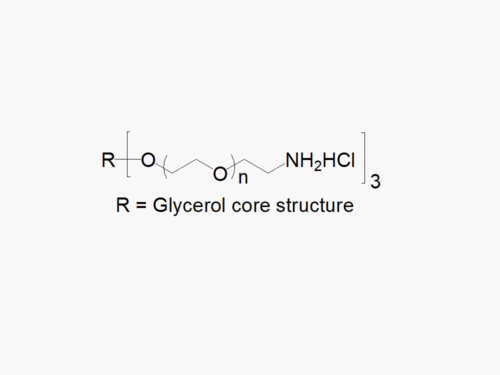
4arm PEG Amine, Free Amine
$70.00 – $560.00
Description
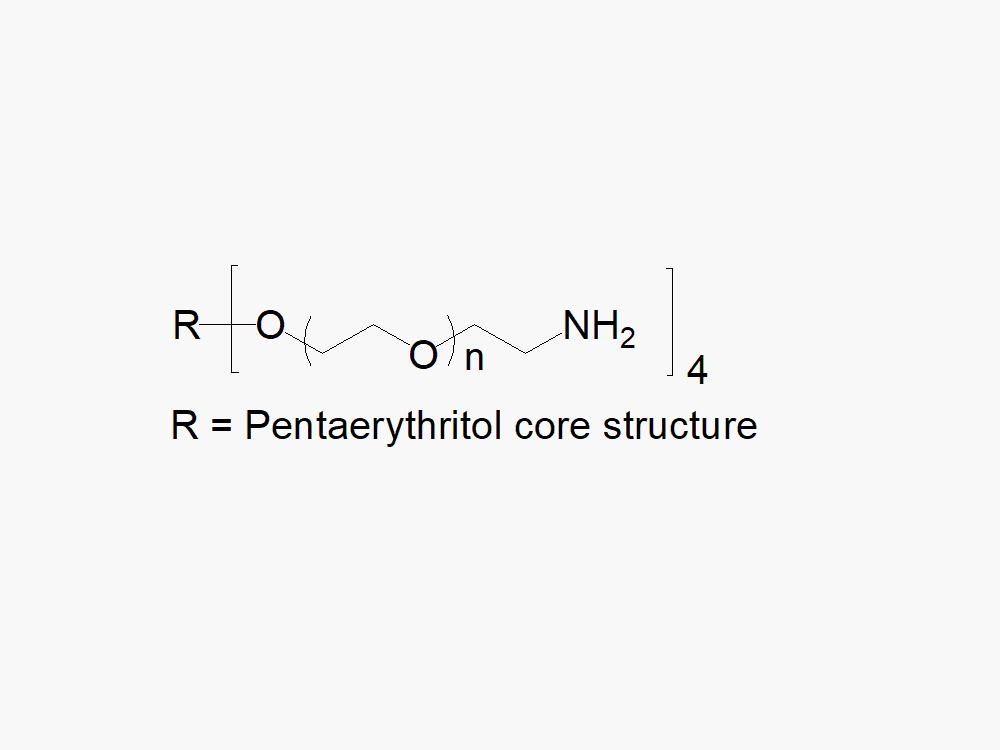
4arm PEG Amine, Free Amine, reagent with superior quality specification of ≥ 95% Substitution.
JenKem Technology’s 4arm PEG Amine derivatives can be cross-linked into PEG hydrogels. PEG hydrogels have a variety of applications in medical devices and regenerative medicine, and are especially of interest for controlled release of drugs, for 2D and 3D cell culture, and for wound sealing and healing. JenKem Technology’s 4 arm PEGs are synthesized by ethoxylation of pentaerythritol. The number of ethylene oxide units in the PEG chain may not be equal for all arms. The total molecular weight reported for the JenKem multi-arm PEGs is the sum of the PEG molecular weights of each arm.
Multi-arm star PEG products with molecular weights, branching, and functional groups not listed in our online catalog may be available by custom synthesis. Please inquire at tech@jenkemusa.com about pricing and availability.
Bulk PEGs and GMP grade PEGs are made-to-order. Please contact us for bulk pricing.
Click here to download the MSDS
References:
1. Zhang, C., et al., Synergic highly effective photothermal-chemotherapy with platinum prodrug linked melanin-like nanoparticles, Artificial cells, nanomedicine, and biotechnology, 2018, pp.1-8.
2. Bachmann, D., et al., Retargeting of UniCAR T cells with an in vivo synthesized target module directed against CD19 positive tumor cells, Oncotarget, 2018, 9(7), p.7487.
3. Ding, Y., et al., Biomimetic soft fibrous hydrogels for contractile and pharmacologically responsive smooth muscle, Acta biomaterialia, 2018.
4. Akbari, S., et al., A versatile microfluidic device for high throughput production of microparticles and cell microencapsulation, Lab on a Chip, 2017.
5. Galvin, O., et al., A sustained release formulation of novel quininib-hyaluronan microneedles inhibits angiogenesis and retinal vascular permeability in vivo. Journal of Controlled Release, 2016, 233:198-207.
6. Mohd Isa, I. L., et al., Hyaluronic Acid Based Hydrogels Attenuate Inflammatory Receptors and Neurotrophins in Interleukin-1beta Induced Inflammation Model of Nucleus Pulposus Cells, Biomacromolecules, 2015, 16 (6), 1714-1725.
7. DeForest, C.A., et al., A photoreversible protein-patterning approach for guiding stem cell fate in three-dimensional gels, Nature Materials, 2015, 14, 523–531.
8. Fan M, Z., et al., Injectable Multi-Arm Poly (ethylene glycol)/Hyaluronic Acid Hydrogels for Adipose Tissue Engineering. Journal of Macromolecular Science, Part A., 2015 , 52(5):345-52.
9. Myers, B.K., et al., The characterization of dendronized poly(ethylene glycol)s and poly(ethylene glycol) multi-arm stars using matrix-assisted laser desorption/ionization time-of-flight mass spectrometry, Analytica Chimica Acta, 2014, 808(0): p. 175-189.
10. Hao, Y., et al., Visible light cured thiol-vinyl hydrogels with tunable degradation for 3D cell culture, Acta Biomaterialia, 2014, 10(1): p. 104-114.
11. Xu, J., E. Feng, and J. Song, Bioorthogonally Cross-Linked Hydrogel Network with Precisely Controlled Disintegration Time over a Broad Range, Journal of the American Chemical Society, 2014, 136(11): p. 4105-4108.
12. McKinnon, D.D., et al., Bis-Aliphatic Hydrazone-Linked Hydrogels Form Most Rapidly at Physiological pH: Identifying the Origin of Hydrogel Properties with Small Molecule Kinetic Studies. Chemistry of Materials, 2014, 26(7): p. 2382-2387.
13. Iemsam-Arng, J., et al., Star Shaped Poly(ethylene glycols) Yield Biocompatible Gene Delivery Systems, Pharmaceutical Nanotechnology, 2014, V. 2:4, pp. 182-195(14).
14. Gandavarapu, N. R., et al., Osteogenic differentiation of human mesenchymal stem cells on α5 integrin binding peptide hydrogels is dependent on substrate elasticity, Biomaterials Science, 2014, 2(3): 352-361.
15. McKinnon, D.D., et al., Synthetic hydrogel platform for three-dimensional culture of embryonic stem cell-derived motor neurons, Biomater. Sci., 2013, 1, 460-469.
16. Maitz, M.F., et al., Bio-responsive polymer hydrogels homeostatically regulate blood coagulation. Nature Commun, 2013. 4.
17. Raza, A., et al., The Influence of Matrix Degradation and Functionality on Cell Survival and Morphogenesis in PEG-Based Hydrogels, Macromolecular Bioscience, 2013, 13(8): 1048-1058.
18. Shih, H., et al., Visible-Light-Mediated Thiol-Ene Hydrogelation Using Eosin-Y as the Only Photoinitiator, Macromolecular Rapid Communications, 2013, 34(3): 269-273.
19. Alge, D. L., et al., Synthetically tractable click hydrogels for three-dimensional cell culture formed using tetrazine–norbornene chemistry, Biomacromolecules, 2013, 14.4 : 949-953.
20. Rane, A.A., Understanding mechanisms by which injectable biomaterials affect cardiac function postmyocardial infarction, UC San Diego, 2012.
21. Tan, H., et al., Injectable nanohybrid scaffold for biopharmaceuticals delivery and soft tissue engineering. Macromolecular rapid communications, 2012, 33(23):2015-22.
22. Ward, J., et al., Amine Functionalization of Collagen Matrices with Multifunctional Polyethylene Glycol Systems, Biomacromolecules, 2010, 11(11) p: 3093–3101.
23. Tan, H., et al., Novel Multi-arm PEG-based Hydrogels for Tissue Engineering, Journal of biomedical materials research Part A., 2010, 92(3):979-987.
Founded in 2001 by experts in PEG synthesis and PEGylation, JenKem Technology specializes exclusively in the development and manufacturing of high quality polyethylene glycol (PEG) products and derivatives, and related custom synthesis and PEGylation services. JenKem Technology is ISO 9001 and ISO 13485 certified, and adheres to ICH Q7A guidelines for GMP manufacture. The production of JenKem® PEGs is back-integrated to in-house polymerization from ethylene oxide, enabling facile traceability for regulated customers. JenKem Technology caters to the PEGylation needs of the pharmaceutical, biotechnology, medical device and diagnostics, and emerging chemical specialty markets, from laboratory scale through large commercial scale.