Experience has shown that siRNA drugs, while potentially very useful, are easily degraded by nucleases in vivo, and their relatively high molecular weight, negative charge and hydrophilicity inhibit their ability to permeate through cell membranes [2, 3].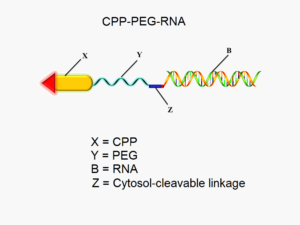
To circumvent these deficiencies, a novel drug delivery system consisting of a cell penetrating peptide (LMWP) linked to activated PEGs (polyethylene glycol polymers) has been developed for siRNA delivery. Several in-stock products are listed below, please contact us for details:
| PEG LMWP Derivative | Packing |
| OPSS PEG3500 LMWP | 5mg/bottle |
| OPSS PEG5000 LMWP | 5mg/bottle, 10mg/bottle |
| OPSS PEG7500 LMWP | 5mg/bottle |
| Maleimide PEG5000 LMWP | 5mg/bottle, 10mg/bottle |
| Maleimide PEG7500 LMWP | 5mg/bottle |
The addition of the PEG improves the drug’s bioavailability by preventing the self-assembly and formation of intra-molecular hairpin structures between the cell penetrating peptide and siRNA [4].
These novel Cell Penetrating Peptide PEG nonviral gene delivery reagents were launched in May 2018 via the “Novel Cell-Penetrating Drug Delivery System for siRNA” poster that was presented by JenKem Technology and Tianjin Medical University at the 2018 TIDES meeting [1].
In vitro studies confirm successful cellular uptake of siRNA and high gene-silencing efficacy using this novel cell penetrating peptide-PEG drug delivery system. LMWP-PEG was first conjugated with FITC labeled anti-EGFP siRNA. Cell uptake studies for LMWP-PEG-RNA conjugate with anti-EGFP siRNA were carried out on MDA-MB-231 cells using a confocal microscope.
The gene silencing down-regulating effect by anti-EGFP siRNA was assessed by laser scanning microscopy using EGFP over-expressed MDA-MB-231-EGFP cells.
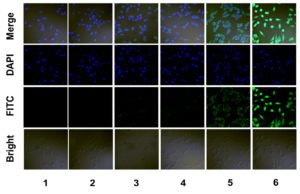
Figure 1. Intracellular uptake study.
(1) PBS (2) anti-EGFP siRNA (3) Lipofecter and anti-GFP siRNA complex (4) LMWP-PEG and anti-EGFP siRNA mixture (molar ratio: 1:1) (5) LMWP and anti-EGFP siRNA mixture (molar ratio: 1:1), and (6) LMWP-PEG-anti-EGFP siRNA conjugate.
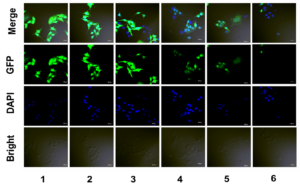
Figure 2. Gene silencing study
(1) PBS, (2) siRNA negative control, (3) anti-EGFP siRNA, (4) Lipofecter and anti-EGFP siRNA complex, (5) LMWP and anti-EGFP siRNA mixture (molar ratio: 1:1), and (6) the LMWP-PEG-anti-EGFP siRNA conjugate.
As seen in Figure 1, anti-EGFP siRNA alone cannot penetrate the cells membrane. The fluorescence intensity of LMWP-PEG-RNA inside the cells is very strong, suggesting that the cellular uptake after conjugation with this novel drug delivery system is significant. As seen in Figure 2, LMWP-PEG-RNA has a high membrane penetrating ability and significantly inhibits target gene expression.
LMWP-PEGs can be employed for gene delivery, and to deliver other drug molecules that cannot penetrate the cell membrane on their own.
Please contact us for a quotation.
Please visit this link for information on PEG reagents for mRNA delivery, such as M-DMG-2000 for LNPs.
References:
1. Zhao, X., et al., Novel Cell-Penetrating Drug Delivery System for siRNA, Poster presentation at 2018 TIDES.
2. Fire, A., et al., Potent and specific genetic interference by double-stranded RNA in Caenorhabditis elegans. Nature. 1998; 391:806-11.
3. Chang, L.C., et al., Low molecular weight protamine (LMWP) as nontoxic heparin/low molecular weight heparin antidote (I): preparation and characterization. AAPS PHARMSCI 2001; 3:7e14.
4. Ye, J., et al., High-Yield Synthesis of Monomeric LMWP(CPP)-siRNA Covalent Conjugate for Effective Cytosolic Delivery of siRNA. Theranostics 2017,7(9):2495-2508.
Founded in 2001 by experts in PEG synthesis and PEGylation, JenKem Technology specializes exclusively in the development and manufacturing of high quality polyethylene glycol (PEG) products and derivatives, and related custom synthesis and PEGylation services. JenKem Technology is ISO 9001 and ISO 13485 certified, and adheres to ICH Q7A guidelines for GMP manufacture. The production of JenKem® PEGs is back-integrated to in-house polymerization from ethylene oxide, enabling facile traceability for regulated customers. JenKem Technology caters to the PEGylation needs of the pharmaceutical, biotechnology, medical device and diagnostics, and emerging chemical specialty markets, from laboratory scale through large commercial scale.
