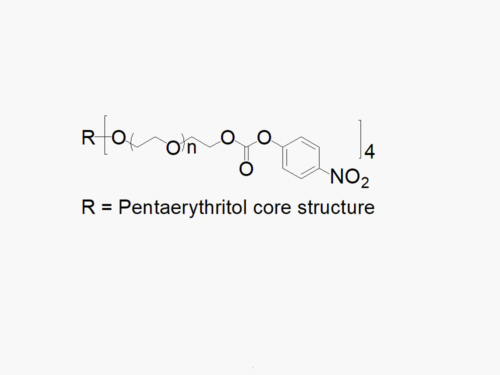
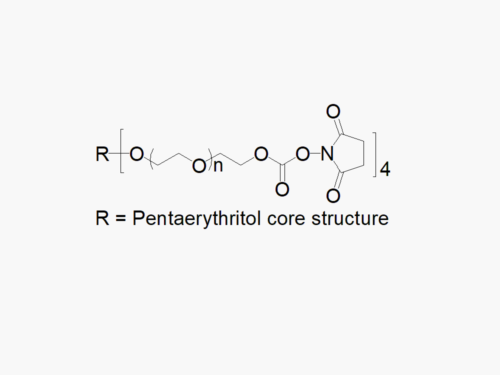
PEG products with additional MW may be made to order, please contact us for details
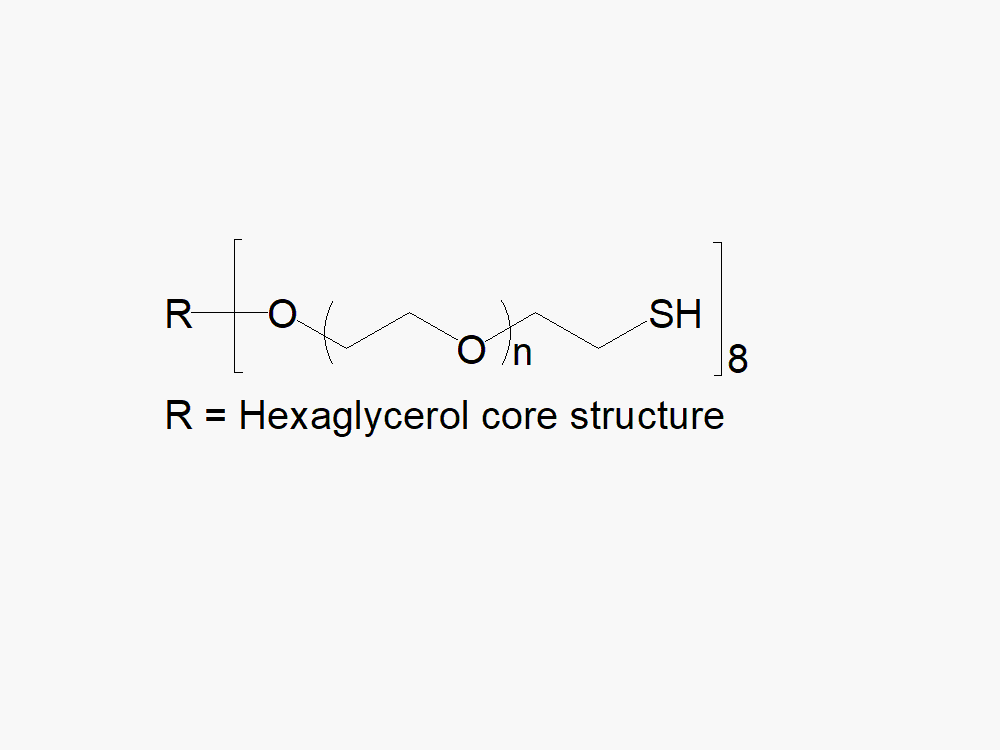
8arm PEG Thiol (hexaglycerol)
$80.00 – $960.00
Description
8arm PEG Thiol (hexaglycerol) with superior quality specification of > 90% Substitution.
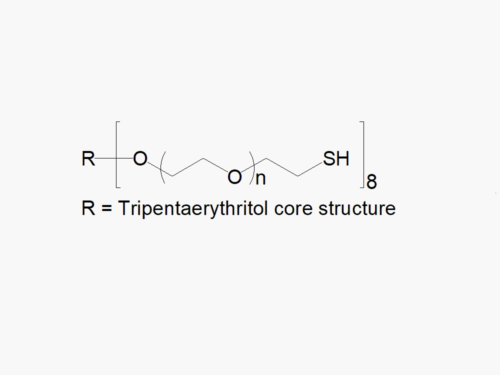
JenKem Technology’s 8arm PEG Thiol (hexaglycerol) HCl salt derivatives can be crosslinked into PEG hydrogels. PEG hydrogels have a variety of applications in medical devices and regenerative medicine, and are especially of interest for controlled release of drugs, for 2D and 3D cell culture, and for wound sealing and healing. JenKem Technology’s 8 arm PEGs are synthesized by ethoxylation of tripentaerythritol (8ARM PEG(TP)) or hexaglycerol (8ARM PEG). The number of ethylene oxide units in the PEG chain may not be equal for all arms. The total molecular weight reported for the JenKem multi-arm PEGs is the sum of the PEG molecular weights of each arm.
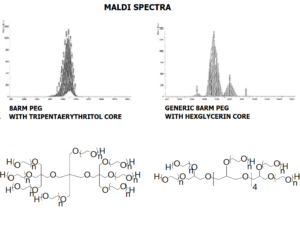
JenKem Technology also provides 8ARM(TP)-PEG hydroxyl raw materials and PEG derivatives with tripentaerythritol core. 8ARM(TP)-PEGs with tripentaerythritol core have a higher purity as evidenced by MALDI compared with the generic 8ARM-PEGs with a hexaglycerin core.
Multi-arm star PEG products with molecular weights, branching, and functional groups not listed in our online catalog may be available by custom synthesis. Please inquire at tech@jenkemusa.com about pricing and availability.
Bulk PEGs and GMP PEGs are made-to-order. Please contact us for bulk pricing.
References:
- Pham-Nguyen, O.V, et al., Complete breakdown of copper-free clickable doxorubicin nanoclusters for real-time tumor proliferation tracking, Chemical Engineering Journal, 468, 2023.
- Su, Q., et al., Facile preparation of a metal-phenolic network-based lymph node targeting nanovaccine for antitumor immunotherapy, Acta Biomaterialia, V. 158, 2023, P. 510-524.
- Zhang, C., et al., Mechanics-driven nuclear localization of YAP can be reversed by N-cadherin ligation in mesenchymal stem cells. Nature Communications. 2021, 12(1):1-3.
- Swaminathan, G., et al., Effect of substrate stiffness on human intestinal enteroids’ infectivity by enteroaggregative Escherichia coli, Acta Biomaterialia, 2021, V. 132.
- Rao, V.V., et al., Rescuing mesenchymal stem cell regenerative properties on hydrogel substrates post serial expansion, Bioengineering & translational medicine, 2019.
- Brown, T.E., et al., Photopolymerized dynamic hydrogels with tunable viscoelastic properties through thioester exchange, Biomaterials, 2018.
- Aziz, A.H., et al., Mechanical characterization of sequentially layered photo-clickable thiol-ene hydrogels, Journal of the Mechanical Behavior of Biomedical Materials, 2017, V. 65, p. 454-465.
- Huynh, C.T., et al., Cytocompatible Catalyst-free Photodegradable Hydrogels for Light-Mediated RNA Release to Induce hMSC Osteogenisis. ACS Biomaterials Science & Engineering, 2017.
- Suma, T., et al., Modulated Fragmentation of Proapoptotic Peptide Nanoparticles Regulates Cytotoxicity. J. Am. Chem. Soc., 2017, 139(11):4009-18.
- Liang, Y., et al., Controlled release of an anthrax toxin-neutralizing antibody from hydrolytically degradable polyethylene glycol hydrogels, Journal of Biomedical Materials Research Part A, 2016, V. 104:1, p. 113–123.
- Lee, S., et al., Effects of the poly(ethylene glycol) hydrogel crosslinking mechanism on protein release, Biomater. Sci., 2016.
- Huynh, C.T., et al., Light-triggered RNA release and induction of hMSC osteogenesis via photodegradable, dual-crosslinked hydrogels. Nanomedicine, 2016.
- Shih, H., et al., Photo-click hydrogels prepared from functionalized cyclodextrin and poly(ethylene glycol) for drug delivery and in situ cell encapsulation, Biomacromolecules, 2015.
- Nguyen, M.K., et al., Sustained localized presentation of RNA interfering molecules from in situ forming hydrogels to guide stem cell osteogenic differentiation. Biomaterials, 2014, 35(24), p. 6278-6286.
- Lee, S., et al., The effects of varying poly(ethylene glycol) hydrogel crosslinking density and the crosslinking mechanism on protein accumulation in three-dimensional hydrogels, Acta Biomaterialia, 2014, 10(10), p. 4167–4174.
Click here to download the MSDS
Founded in 2001 by experts in PEG synthesis and PEGylation, JenKem Technology specializes exclusively in the development and manufacturing of high quality polyethylene glycol (PEG) products and derivatives, and related custom synthesis and PEGylation services. JenKem Technology is ISO 9001 and ISO 13485 certified, and adheres to ICH Q7A guidelines for GMP manufacture. The production of JenKem® PEGs is back-integrated to in-house polymerization from ethylene oxide, enabling facile traceability for regulated customers. JenKem Technology caters to the PEGylation needs of the pharmaceutical, biotechnology, medical device and diagnostics, and emerging chemical specialty markets, from laboratory scale through large commercial scale.