PEG products with additional MW may be made to order, please contact us for details
4arm PEG Maleimide
$130.00 – $1,120.00
Description
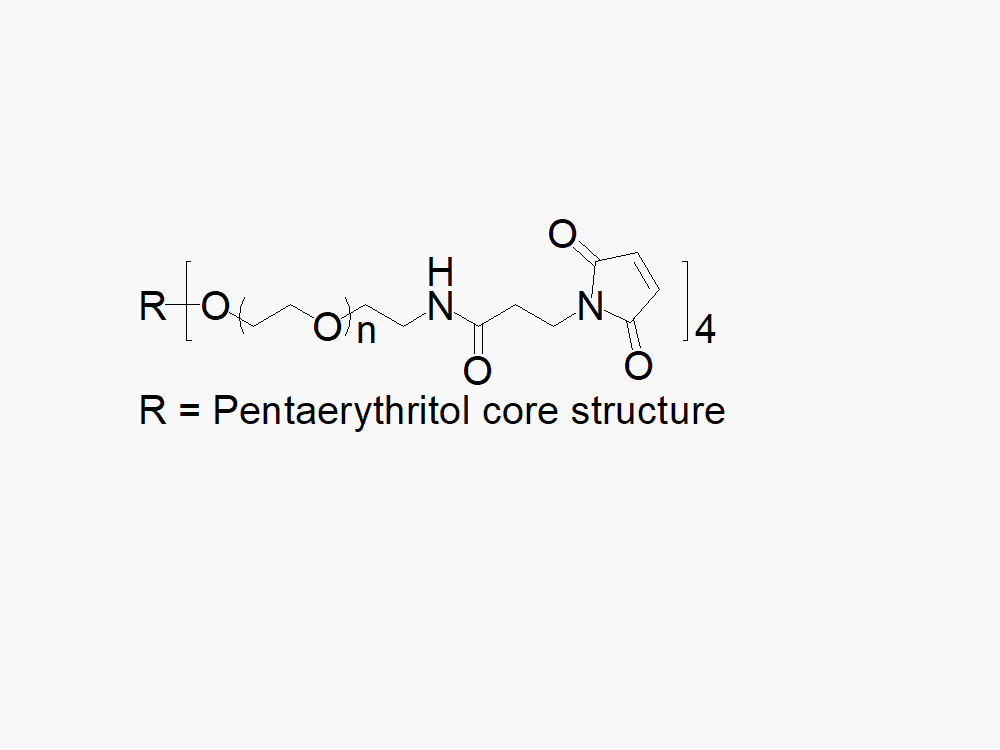
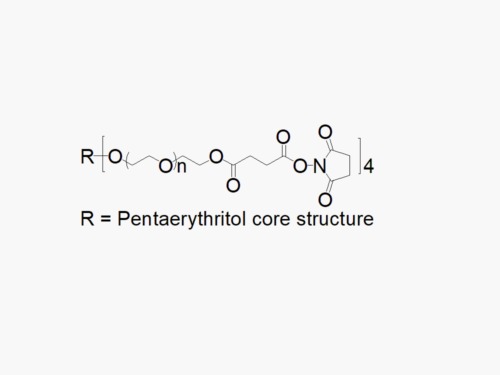
4arm PEG Maleimide reagent with superior quality specification of > 90% Substitution.
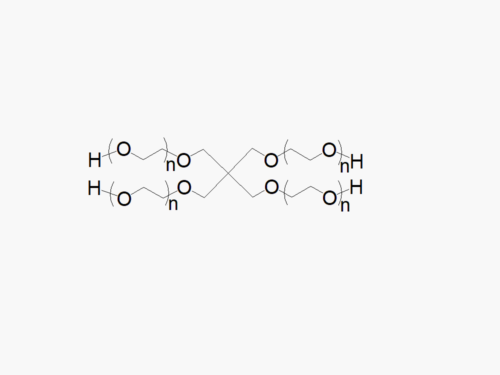
JenKem Technology’s 4arm PEG Maleimide derivatives can be cross-linked into PEG hydrogels. PEG hydrogels have a variety of applications in medical devices and regenerative medicine, and are especially of interest for controlled release of drugs, for 2D and 3D cell culture, and for wound sealing and healing. JenKem Technology’s 4arm PEGs are synthesized by ethoxylation of pentaerythritol. The number of ethylene oxide units in the PEG chain may not be equal for all arms. The total molecular weight reported for the JenKem multi-arm PEGs is the sum of the PEG molecular weights of each arm.
4ARM-MAL-10K, 4ARM-MAL-20K and 4ARM-MAL-40K can be ordered online. 4arm MAL PEG products with molecular weights, branching, and functional groups not listed in our online catalog may be available by custom synthesis. Please inquire at tech@jenkemusa.com about pricing and availability.
Bulk PEGs and GMP grade PEGs are made-to-order. Please contact us for bulk pricing.
Click here to download the MSDS
References:
- Martin, K. E., et al., Hydrolytic hydrogels tune mesenchymal stem cell persistence and immunomodulation for enhanced diabetic cutaneous wound healing, Biomaterials, 301, 2023.
- Ilochonwu, B. C., et al., Thermo-responsive Diels-Alder stabilized hydrogels for ocular drug delivery of a corticosteroid and an anti-VEGF fab fragment, Journal of Controlled Release, 361, 2023.
- Xu, Y., et al., A self-assembled dynamic extracellular matrix-like hydrogel system with multi-scale structures for cell bioengineering applications, Acta Biomaterialia, V. 162, 2023, P. 211-225.
- Bock N, et al., GelMA, Click-Chemistry Gelatin and Bioprinted Polyethylene Glycol-Based Hydrogels as 3D Ex Vivo Drug Testing Platforms for Patient-Derived Breast Cancer Organoids. Pharmaceutics. 2023; 15(1):261.
- Wu, N., et al., Correlation of Bulk Degradation and Molecular Release from Enzymatically Degradable Polymeric Hydrogels. Biomacromolecules. 2021.
- Cheng, L., et al., Bioresponsive micro-to-nano albumin-based systems for targeted drug delivery against complex fungal infections. Acta Pharmaceutica Sinica B. 2021
- Liu, S, et al., Injectable and Degradable PEG Hydrogel with Antibacterial Performance for Promoting Wound Healing. ACS Applied Bio Materials. 2021, 4(3):2769-80.
- Guo, R, et al., Anticalcification Potential of POSS-PEG Hybrid Hydrogel as a Scaffold Material for the Development of Synthetic Heart Valve Leaflets. ACS Applied Bio Materials. 2021, 4(3):2534-43.
- Kim, J, et al., In Situ Crosslinked Hydrogel Depot for Sustained Antibody Release Improves Immune Checkpoint Blockade Cancer Immunotherapy. Nanomaterials. 2021, 11(2):471.
- Scott, R. A., et al., Substrate stiffness directs the phenotype and polarization state of cord blood derived macrophages, Acta Biomaterialia, 2021, V. 122, P. 220-235.
- Wang, J., et al., An injectable PEG hydrogel controlling neurotrophin-3 release by affinity peptides, Journal of Controlled Release, 2021, 330, P. 575-586.
- Schirmer, L., et al., Glycosaminoglycan-based hydrogels with programmable host reactions, Biomaterials, 2020, V. 228.
- Kwon, M. Y., et al., Influence of hyaluronic acid modification on CD44 binding towards the design of 4hydrogel biomaterials, Biomaterials, 2019, 222.
- Li, H., et al., Synthesis of thiol-terminated PEG-functionalized POSS cross-linkers and fabrication of high-strength and hydrolytic degradable hybrid hydrogels in aqueous phase, European Polymer Journal, 2019, 116:74-83.
- Atallah, P., et al., Charge-tuning of glycosaminoglycan-based hydrogels to program cytokine sequestration, Faraday Discussions, 2019.
- Dai, J., et al., Modifying decellularized aortic valve scaffolds with stromal cell-derived factor-1α loaded proteolytically degradable hydrogel for recellularization and remodeling, Acta biomaterialia, 2019.
- Tunn, I., et al., Bioinspired Histidine–Zn2+ Coordination for Tuning the Mechanical Properties of Self-Healing Coiled Coil Cross-Linked Hydrogels, Biomimetics, 2019, 4(1):25.
- Jansen, L.E., et al., Control of thiol-maleimide reaction kinetics in PEG hydrogel networks, Acta Biomaterialia, 2018, V. 70, P. 120-128.
- Schweikle, M., et al.,. Injectable synthetic hydrogel for bone regeneration: Physicochemical characterisation of a high and a low pH gelling system, Materials Science and Engineering: C, 2018, 90, pp.67-76.
- Brooks, E.A., et al., Complementary, Semi-automated Methods for Creating Multi-dimensional, PEG-based Biomaterials, ACS Biomaterials Science & Engineering, 2018.
- Matsumura, K., et al., Urokinase injection-triggered clearance enhancement of a 4-arm PEG-conjugated 64 Cu-bombesin analog tetramer: A novel approach for the improvement of PET imaging contrast, International journal of pharmaceutics, 2018.
- Yang, T., et al., Superparamagnetic colloidal chains prepared via Michael-addition, Colloids and Surfaces A: Physicochemical and Engineering Aspects, 2018, V. 540, P. 23-28.
- Shen, J., et al., Hydrolytically degradable POSS-PEG hybrid hydrogels prepared in aqueous phase with tunable mechanical properties, swelling ratio and degradation rate, Reactive and Functional Polymers, 2018, V. 123, P. 91-96.
- Day, J.R., et al., The impact of functional groups of poly(ethylene glycol) macromers on the physical properties of photo-polymerized hydrogels and the local inflammatory response in the host, Acta Biomaterialia, 2018, Vol. 67, P. 42-52.
- Tondera, C., et al., In Vivo Examination of an Injectable Hydrogel System Crosslinked by Peptide–Oligosaccharide Interaction in Immunocompetent Nude Mice, Advanced Functional Materials, 2017, 27(15).
- Maitz, M.F., et al., Adaptive release of heparin from anticoagulant hydrogels triggered by different blood coagulation factors, Biomaterials, 2017, 135:53-61.
- Robinson, K.G., et al., Reduced Arterial Elasticity due to Surgical Skeletonization is Ameliorated by Abluminal PEG Hydrogel, Bioengineering & Translational Medicine, 2017.
- Bas, O., et al., Biofabricated soft network composites for cartilage tissue engineering. Biofabrication, 2017.
- Nowak, M., et al., Modular GAG-matrices to promote mammary epithelial morphogenesis in vitro, Biomaterials 2017, 112, p. 20-30.
- Hesse, E., et al., Peptide‐functionalized starPEG/heparin Hydrogels Direct Mitogenicity, Cell Morphology and Cartilage Matrix Distribution in vitro and in vivo. Journal of Tissue Engineering and Regenerative Medicine, 2017.
- Gencoglu, M.F., et al., Comparative study of multicellular tumor spheroid formation methods and implications for drug screening. ACS Biomaterials Science & Engineering, 2017.
- Skoumal, M.J, Localized Tolerance and Development of an Alternative Transplant Site to Treat Type 1 Diabetes, University of Michigan, 2017.
- Wu, F., et al., A novel synthetic microfiber with controllable size for cell encapsulation and culture. Journal of Materials Chemistry B, 2016, 4(14):2455-65.
- Taubenberger, A.V., et al., 3D extracellular matrix interactions modulate tumour cell growth, invasion and angiogenesis in engineered tumour microenvironments, Acta biomaterialia, 2016.
- Darling, N.J., et al., Controlling the kinetics of thiol-maleimide Michael-type addition gelation kinetics for the generation of homogenous poly (ethylene glycol) hydrogels, Biomaterials, 2016.
- Danmark, S., et al., Tailoring Supramolecular Peptide-Poly (ethylene glycol) Hydrogels by Coiled Coil Self-Assembly and Self-Sorting. Biomacromolecules, 2016.
- Rios, P.D., et al., Mold‐casted non‐degradable, islet macro‐encapsulating hydrogel devices for restoration of normoglycemia in diabetic mice. Biotechnology and bioengineering, 2016.
- Wang, J.J, et al., Biomimetic synthesis of platelet-shaped hydroxyapatite mesocrystals in a collagen mimetic peptide–PEG hybrid hydrogel, Materials Letters, 2015, 159, P. 150-153.
- Mahadevaiah, S., et al., Decreasing matrix modulus of PEG hydrogels induces a vascular phenotype in human cord blood stem cells, Biomaterials, 2015, 62, p. 24-34.
- Griffin, D. R., et al., Hybrid Photopatterned Enzymatic Reaction (HyPER) for In situ Cell Manipulation, Chembiochem : a European journal of chemical biology, 2014, 15(2): 233-242.
- Liang, Y., et al., Multifunctional lipid-coated polymer nanogels crosslinked by photo-triggered Michael-type addition, Polym. Chem., 2014, 5, 1728-1736.
- Maitz, M.F., et al., Bio-responsive polymer hydrogels homeostatically regulate blood coagulation, Nat Commun, 2013, 4.
- Robinson, K. G., et al., Differential effects of substrate modulus on human vascular endothelial, smooth muscle, and fibroblastic cells, Journal of Biomedical Materials Research, 2012, 100(5): 1356-1367.
- Lu, H.D., et al., Injectable shear-thinning hydrogels engineered with a self-assembling Dock-and-Lock mechanism, Biomaterials, 2012, 33(7): p. 2145-2153.
- Soon, A.S.C., et al., Modulation of fibrin matrix properties via knob:hole affinity interactions using peptide–PEG conjugates, Biomaterials, 2011, 32:19, P. 4406-4414.
- Nie, T., et al., Production of heparin-containing hydrogels for modulating cell responses, Acta Biomaterialia, 2009, 5(3), p: 865-875.
Founded in 2001 by experts in PEG synthesis and PEGylation, JenKem Technology specializes exclusively in the development and manufacturing of high quality polyethylene glycol (PEG) products and derivatives, and related custom synthesis and PEGylation services. JenKem Technology is ISO 9001 and ISO 13485 certified, and adheres to ICH Q7A guidelines for GMP manufacture. The production of JenKem® PEGs is back-integrated to in-house polymerization from ethylene oxide, enabling facile traceability for regulated customers. JenKem Technology caters to the PEGylation needs of the pharmaceutical, biotechnology, medical device and diagnostics, and emerging chemical specialty markets, from laboratory scale through large commercial scale.