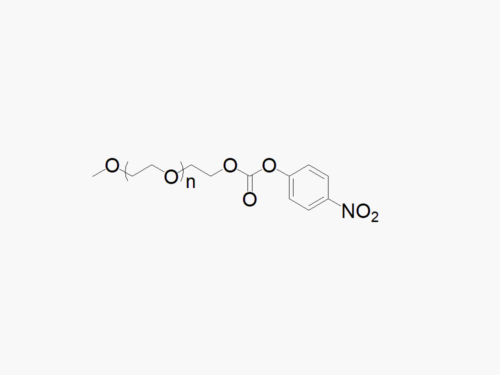
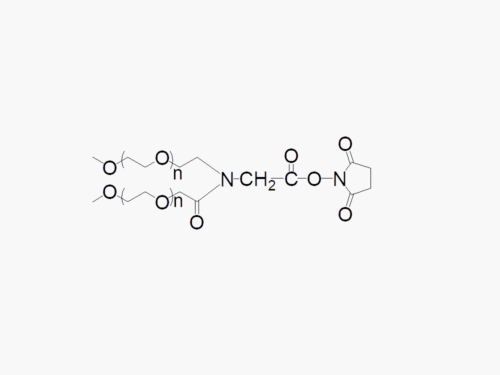
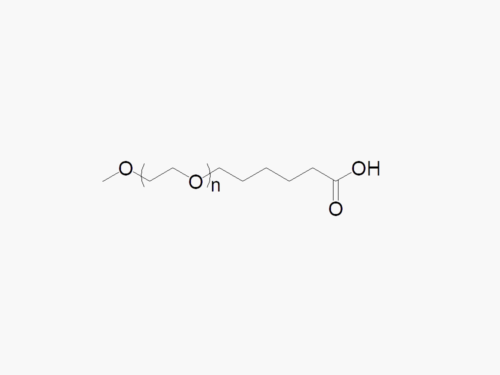
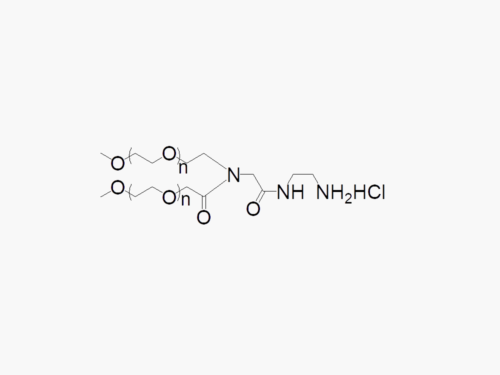
These products are made to order. Please contact us to learn more.
PEG Cyclooctyne Derivatives
Description
Click reactions are quantitative, rapid, have high efficiency, stereo selectivity, and few or no by-products under mild reaction conditions. The traditional click reaction refers to azide and 1,3-dipole ring addition reaction with the catalyst copper (I). As known, copper (I) is cytotoxic and it can damage red blood cells which makes the traditional click reaction limited in its use in biomedical applications.
As early as 1961, the publication “Zur Existenz Niedergliedriger Cycloalkine, I and II” mentioned the phenyl azide and cycloalkines reacting without the need for a copper (I) catalyst. However, most of the cyclooctyne reagents do not have good water solubility, which limits their applications in biomedicine.
JenKem Technology patented a series of polyethylene glycol-cyclooctyne derivatives which react with azide without copper(I) catalyst, thus making the post-processing very simple and eliminating the cytotoxicity issues of the traditional click chemistry. JenKem® PEG-cyclooctynes have good water solubility and biocompatibility. Polyethylene glycol derivatives are widely used in polypeptide, protein and drug delivery to prolong half-life, and reduce immunogenicity and toxicity; either in combination with a drug molecule via PEGylation, or as part of hydrogels.
Advantages of Novel PEG Cyclooctyne Derivatives for Click Chemistry:
- React with azide without cytotoxic copper (I) catalyst
- No cytotoxicity issues like those associated with traditional click chemistry
- Good water solubility and biocompatibility
- Applications in drug delivery to prolong half-life, and reduce drug immunogenicity and toxicity
- Multiarm PEG-cyclooctynes available for hydrogel formation
These products are made to order, please contact us for details.
JenKem Technology also has extensive capability to provide high quality PEG Dibenzocyclooctyne products in large kilogram-scale, in both non-GMP and GMP grade, at affordable prices. Below are several examples of PEG DBCO products manufactured by JenKem Technology. PEG and monodisperse PEG DBCO derivatives with different branching, MW, and functionalities may be available by custom synthesis.
Methoxy PEG DBCO Products
| Item Number | Item Description |
| M-DBCO-2000 | Methoxy PEG Dibenzocyclooctyne, MW 2000 |
| M-DBCO-3500 | Methoxy PEG Dibenzocyclooctyne, MW 3500 |
| M-DBCO-5000 | Methoxy PEG Dibenzocyclooctyne, MW 5000 |
| M-DBCO-10K | Methoxy PEG Dibenzocyclooctyne, MW 10000 |
| M-DBCO-20K | Methoxy PEG Dibenzocyclooctyne, MW 20000 |
| M-DBCO-30K | Methoxy PEG Dibenzocyclooctyne, MW 30000 |
| M-DBCO-40K | Methoxy PEG Dibenzocyclooctyne, MW 40000 |
Y-Shaped and U-Shaped PEG DBCO Products
| Item Number | Item Description |
| Y-DBCO-40K | Y-shape (MPEG20K)2 Dibenzocyclooctyne, MW 40000 |
| MPEG2-DBCO-40K | MPEG2 Dibenzocyclooctyne, MW 40000 |
Homobifunctional PEG DBCO Products
| Item Number | Item Description |
| DBCO-PEG2000-DBCO | PEG (Dibenzocyclooctyne)2, MW 2000 |
| DBCO-PEG5000-DBCO | PEG (Dibenzocyclooctyne)2, MW 5000 |
| DBCO-PEG10K-DBCO | PEG (Dibenzocyclooctyne)2, MW 10000 |
Multi-arm PEG DBCO Products
| Item Number | Item Description |
| 4ARM-DBCO-10K | 4arm PEG Dibenzocyclooctyne (pentaerythritol), MW 10000 |
| 4ARM-DBCO-20K | 4arm PEG Dibenzocyclooctyne (pentaerythritol), MW 20000 |
| 8ARM(TP)-DBCO-10K | 8arm PEG Dibenzocyclooctyne (tripentaerythritol), MW 10000 |
| 8ARM(TP)-DBCO-20K | 8arm PEG Dibenzocyclooctyne (tripentaerythritol), MW 20000 |
| 8ARM(TP)-DBCO-40K | 8arm PEG Dibenzocyclooctyne (tripentaerythritol), MW 40000 |
Heterobifunctional PEG DBCO Products
| Item Number | Item Description |
| DBCO-PEG2000-SCM | Dibenzocyclooctyne PEG Succinimidyl Carboxymethyl Ester, MW 2000 |
| DBCO-PEG3500-SCM | Dibenzocyclooctyne PEG Succinimidyl Carboxymethyl Ester, MW 3500 |
| DBCO-PEG5000-SCM | Dibenzocyclooctyne PEG Succinimidyl Carboxymethyl Ester, MW 5000 |
| DBCO-PEG2000-NH2 | Dibenzocyclooctyne PEG Amine, Free amine, MW 2000 |
| DBCO-PEG3500-NH2 | Dibenzocyclooctyne PEG Amine, Free amine, MW 3500 |
| DBCO-PEG5000-NH2 | Dibenzocyclooctyne PEG Amine, Free amine, MW 5000 |
| DBCO-PEG2000-MAL | Dibenzocyclooctyne PEG Maleimide, MW 2000 |
| DBCO-PEG3500-MAL | Dibenzocyclooctyne PEG Maleimide, MW 3500 |
| DBCO-PEG5000-MAL | Dibenzocyclooctyne PEG Maleimide, MW 5000 |
| DBCO-PEG2000-OH | Dibenzocyclooctyne PEG Hydroxyl, MW 2000 |
| DBCO-PEG3500-OH | Dibenzocyclooctyne PEG Hydroxyl, MW 3500 |
| DBCO-PEG5000-OH | Dibenzocyclooctyne PEG Hydroxyl, MW 5000 |
| DBCO-PEG2000-OPSS | Dibenzocyclooctyne PEG OPSS, MW 2000 |
| DBCO-PEG3500-OPSS | Dibenzocyclooctyne PEG OPSS, MW 3500 |
| DBCO-PEG5000-OPSS | Dibenzocyclooctyne PEG OPSS, MW 5000 |
Monodisperse PEG DBCO Linkers for ADCs
| Product Name | Molecular Weight |
| DBCO-PEG2-PA | 464.5 |
| DBCO-PEG3-PA | 508.6 |
| DBCO-PEG4-PA | 552.6 |
| DBCO-PEG6-PA | 640.7 |
| DBCO-PEG8-PA | 728.8 |
| DBCO-PEG10-PA | 816.9 |
| DBCO-PEG12-PA | 905.0 |
| DBCO-PEG24-PA | 1433.7 |
| DBCO-PEG8-CM | 714.8 |
| DBCO-PEG12-CM | 891.0 |
| DBCO-PEG24-CM | 1419.7 |
| DBCO-PEG8-NH2 | 655.8 |
| DBCO-PEG12-NH2 | 832.0 |
| DBCO-PEG24-NH2 | 1360.6 |
| DBCO-PEG4-VC-PAB | 914.1 |
| DBCO-PEG6-VC-PAB | 1002.2 |
| DBCO-PEG8-VC-PAB | 1090.3 |
| DBCO-PEG12-VC-PAB | 1266.5 |
| DBCO-PEG24-VC-PAB | 1795.1 |
| DBCO-PEG4-VA-PAB | 828.0 |
| DBCO-PEG6-VA-PAB | 916.1 |
| DBCO-PEG8-VA-PAB | 1004.2 |
| DBCO-PEG12-VA-PAB | 1180.4 |
| DBCO-PEG24-VA-PAB | 1709.0 |
JenKem Technology offers custom manufacture for other PEG products for copper-free click chemistry not listed here, please contact us with your custom requests.
Please contact us at sales@jenkemusa.com for a quotation and more information about these PEG products for Copper-Free Click Chemistry.
JenKem Technology specializes in the development and manufacture of high-quality polyethylene glycol (PEG) derivatives, and related custom synthesis and PEGylation services. JenKem Technology is ISO 9001 and ISO 13485 certified and adheres to ICH Q7 guidelines for GMP manufacture. The production of JenKem® PEGs is back integrated to in-house polymerization from ethylene oxide to ensure batch to batch consistency. JenKem Technology caters to the PEGylation needs of the pharmaceutical, biotechnology, medical device and diagnostics, and emerging chemical specialty markets, from laboratory scale through large commercial scale.